Anvisa Approves Leqembi, New Alzheimer’s Drug That Works on the Disease’s Cause and Promises to Slow Its Progression.
The Anvisa approved, on December 22, 2025, a new medication for the treatment of Alzheimer’s in Brazil. It is Leqembi, indicated for people with mild dementia caused by the disease, which works directly in the brain by removing plaques associated with the progression of the condition.
The approval marks a historic advance, as it is the first therapy authorized in the country to focus on the disease’s mechanism, rather than just relieving symptoms.
The decision from Anvisa comes in a context of high demand for new therapeutic alternatives. According to data from the Ministry of Health, over one million Brazilians currently live with Alzheimer’s, a neurodegenerative condition that compromises memory, reasoning, and autonomy.
-
A bright yellow mushroom imported from Asia escaped from a cultivation farm in the United States in 2010 and is now aggressively spreading across 25 states, destroying entire communities of native fungi in American forests.

-
Buried under China, a colossal machine weighing 20,000 tons began operating as the largest neutrino detector on the planet and, in just 59 days, surpassed results that science took half a century to achieve.

-
Iran enters the center of a climate alert after the war emitted 5.6 million tons of CO2 in two weeks, surpassing the annual pollution of entire countries.

-
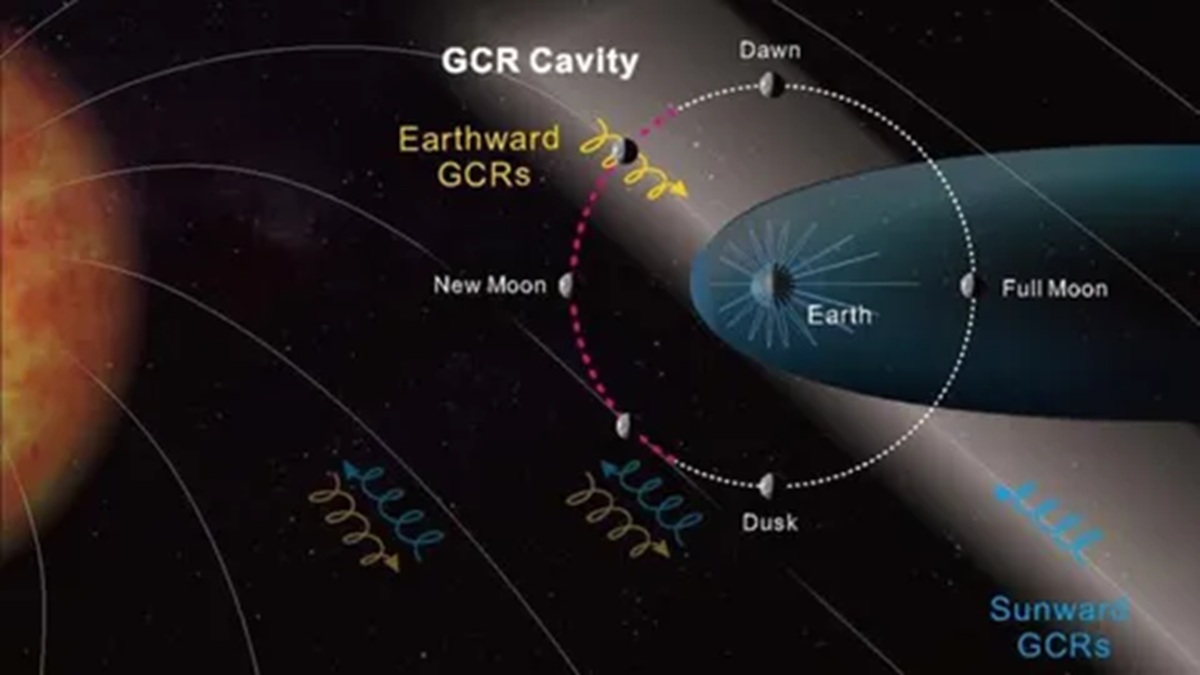
Lunar exploration may change after a Chinese probe finds a “cavity” of radiation that reduces the impact on astronauts by up to 20% at certain times on the Moon.
So far, the available treatments have only acted on clinical manifestations without directly interfering with the evolution of the disease.
What Is Leqembi and How Does the Medication Work on Alzheimer’s
Leqembi is a medication produced from the monoclonal antibody lecanemab. It was developed to recognize and remove beta-amyloid plaques that accumulate in the brains of people with Alzheimer’s, considered one of the main biological markers of the disease.
This antibody works similarly to the body’s natural antibodies, which fight viruses and bacteria.
In the case of Leqembi, it activates the immune system to “clean” the excess beta-amyloid present in the brain tissue. Thus, it aims to reduce the impact of this toxic substance on neurons.
The medication is administered through intravenous infusion and is recommended exclusively for patients in the early stages of Alzheimer’s, when there is already mild cognitive impairment, but there is still a greater potential for therapeutic response.
Why the Approval from Anvisa Is Considered a Milestone
The release of Leqembi by Anvisa represents a significant change in the treatment of Alzheimer’s in Brazil.
For the first time, a medication approved in the country aims to slow the process of brain destruction, intervening in the progression of the disease.
Experts emphasize that, although Leqembi is not a cure, it inaugurates a new therapeutic approach.
Instead of merely alleviating symptoms, the strategy is to act on the biological basis of Alzheimer’s, something that has remained out of reach of medicine for decades.
In addition, the Brazilian approval follows the international endorsement. Since 2023, the medication has been authorized and marketed in the United States, after a rigorous review by the U.S. regulatory agency.
Clinical Study Showed Reduction in Cognitive Decline
The efficacy of Leqembi was demonstrated in a study published in 2022 in the New England Journal of Medicine, one of the most respected scientific journals in the world. The research involved 1,795 volunteers with early-stage Alzheimer’s.
During the study, participants received lecanemab infusions every two weeks. After 18 months of treatment, the results indicated a reduction in cognitive-functional decline, suggesting a slower progression of the disease compared to the group that did not receive the medication.
These data were crucial for the approval of Leqembi by different regulatory agencies, including now Anvisa.
From Support Treatment to Therapies That Attempt to Curb the Disease
For many years, Alzheimer’s was treated only with supportive measures. Until the 1970s, it was known that the disease was associated with brain atrophy and the abnormal accumulation of proteins like tau and beta-amyloid.
However, the available treatments were limited to vitamins, lifestyle changes, and memory stimulants, with no proven efficacy.
With advances in science, researchers began to understand the origins of Alzheimer’s, paving the way for targeted therapies.
Leqembi emerges precisely in this context of evolving knowledge, focusing directly on one of the central mechanisms of the disease.
“This represents a new line of research and a door that opens up opportunities to intervene in the evolution of the disease,” explains Helder Picarelli, a neurosurgeon at the Hospital das Clínicas of the Medical School of USP.
Expectations, Costs, and Caution from Specialists
Despite the enthusiasm surrounding the medication, experts urge caution. Helder Picarelli himself emphasizes that Leqembi is still a relatively new therapy and that more data will be necessary to assess its long-term impact.
“This type of therapy uses a specific antibody; it has changed treatment, but it is still new. I think we need to wait a bit longer to reach a definitive conclusion. I’m not sure if the high cost and the risks outweigh the benefits obtained from this type of medication,” he states.
Thus, the approval of Leqembi by Anvisa represents a concrete advancement in the fight against Alzheimer’s, but also opens a new debate about access, cost, and the real effectiveness of this medication in addressing one of the most daunting diseases of aging.

 Português
Português  Inglês
Inglês  Espanhol
Espanhol 





-

-

-

5 pessoas reagiram a isso.