New Determinations Published In The Official Gazette Affect The Marketing Of Food And Cosmetics Across The Country, Aiming At Consumer Safety.
The National Health Surveillance Agency (Anvisa) issued new resolutions that directly impact the consumer market. The agency prohibited the sale of the children’s food “PF da Nina”, ordered the recall of a batch of Ubon hot sauce, and suspended a body cream “Adeus”. The measures were officially announced last Thursday (28).
Ubon Hot Sauce: A Secret And Dangerous Ingredient
Anvisa determined the suspension of the marketing of the batch 4512823 of the extra hot sauce from the Ubon brand. The decision came after the detection of sulfur dioxide in its composition, a substance that was not declared on the label.
According to the agency, the presence of sulfur dioxide in food, when not disclosed, poses a risk. The chemical compound can cause allergic reactions in people who are sensitive to it.
-
Brazilian city neighboring Paraguay becomes a ‘magnet’ for people seeking quality of life and high standards with exceptional growth and commutes of up to 15 minutes.

-
A rare storm transformed part of the Gulf into an unlikely scene of hail, extreme rain, and tornado risk in the middle of the desert, with volumes that exceeded the annual average in a single day.

-
Brazil catches the world’s attention with ships powered by ethanol and biodiesel that have lower emissions, an unprecedented certification with advantages in chartering, and a revolution in cabotage.

-
A couple who only has Sundays free has built a hand-dug pool, a hamburger joint, a dance hall, and a playground in their own home without hiring a mason, and even with breaks due to accidents and the pandemic, the work has never truly stopped.

Children’s Food “PF da Nina”: Prohibited For Lack Of License And Hygiene
The products “PF da Nina”, which include children’s and transitional foods for infants, have been prohibited throughout Brazil. Anvisa also ordered the seizure of units that are being marketed.
The measure was taken based on a Sanitary Inspection Report that proved the responsible company was producing the items without the proper sanitary license. Furthermore, the manufacturer did not comply with Good Manufacturing Practices, mandatory requirements for food production, especially those intended for children.
Body Cream “Adeus”: The False Promise That Led To Suspension
Another affected product was the multifunctional cream “Adeus”, from JSA Indústria de Cosméticos Ltda. Anvisa suspended the marketing, distribution, manufacturing, advertising, and use of the item.
The agency pointed out that the product, although registered as a cosmetic, presented pharmacological characteristics. Its treatment and cure promises are not allowed for this category of product, which led to its suspension.

 Portuguese
Portuguese  English
English  Spanish
Spanish 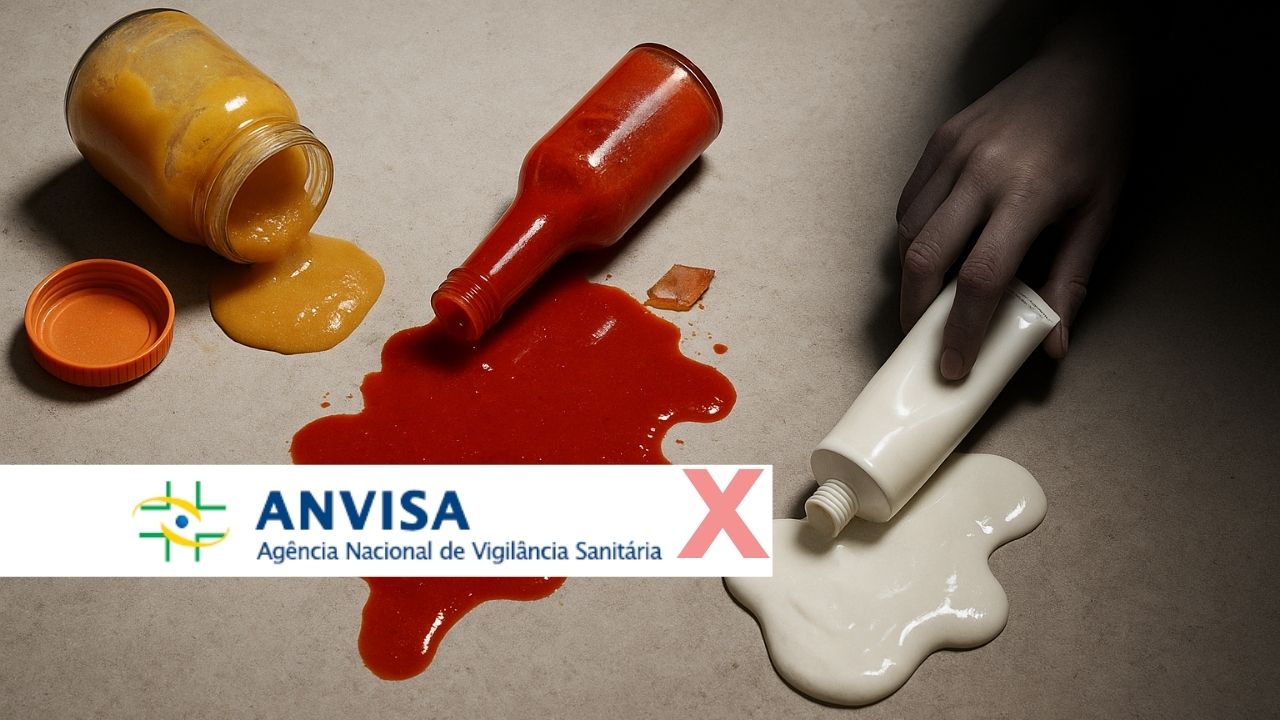






-

Uma pessoa reagiu a isso.