An Experimental Protein Tested by a UFRJ Team Caught the Attention of Researchers and Patients After Reports of Movement Recovery in Severe Spinal Injuries Amid Regulatory Steps and Results Released by Institutions and Reports.
A study led by biologist Tatiana Coelho Sampaio from the Federal University of Rio de Janeiro (UFRJ) tested an experimental protein called poly-Laminin on people with severe spinal cord injury and recorded movement recovery in some participants, according to reports released by institutional channels and reports.
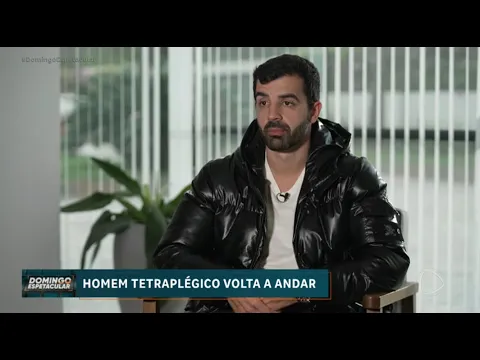
The most cited case in these publications is that of Bruno Drummond de Freitas, who, according to what was published by funding agencies and the university itself, started walking again after receiving the substance.
According to these sources, the treatment remains in the experimental phase and relies on regulatory steps and clinical research to advance.
-
Motorola launched the Signature with a gold seal from DxOMark, tying with the iPhone 17 Pro in camera performance, Snapdragon 8 Gen 5 that surpassed 3 million in benchmarks, and a zoom that impresses even at night.

-
Satellites reveal beneath the Sahara a giant river buried for thousands of kilometers: study shows that the largest hot desert on the planet was once traversed by a river system comparable to the largest on Earth.

-
Scientists have captured something never seen in space: newly born stars are creating gigantic rings of light a thousand times larger than the distance between the Earth and the Sun, and this changes everything we knew about stellar birth.

-
Geologists find traces of a continent that disappeared 155 million years ago after separating from Australia and reveal that it did not sink, but broke into fragments scattered across Southeast Asia.
Therefore, there is no indication of wide availability for use in health services.
Spinal cord injuries can cause loss of motor and sensory function by interrupting communication between the brain and the body.
In severe cases, recovery is usually limited and varies according to the type and extent of trauma, according to medical literature and clinical practice described in rehabilitation materials.
In this context, the proposal of poly-Laminin is to act at the site of the trauma to promote the reorganization of neural connections, as public materials associated with the project describe.
The approach, according to institutional reports, includes application during surgical procedures and rehabilitation monitoring.
Tatiana Coelho Sampaio, UFRJ and the Research on Spinal Cord
Tatiana Sampaio heads the Laboratory of Extracellular Matrix Biology at the Institute of Biomedical Sciences at UFRJ and has studied for decades the role of extracellular matrix proteins in the growth and communication between neurons, according to institutional descriptions.
CAPES states in materials on the subject that the research line began in the late 1990s and evolved into a partnership with the pharmaceutical company Cristália, responsible for producing the compound on a larger scale for studies.
The same publication mentions investment reported by the company and states that UFRJ and Cristália signed a co-ownership agreement in 2021, in addition to mentioning the existence of a patent related to the development of poly-Laminin.
These points are presented as part of the path to enable production under adequate conditions for human research.
Throughout 2025, reports began to highlight preliminary results released by the team and the progress of the processing for clinical studies.
In January 2026, the UFRJ Faculty Association (Adufrj) reported that Anvisa authorized the start of a first phase of clinical study in humans aimed at assessing safety and possible adverse effects.
What Is Poly-Laminin and How Is the Protein Applied
Poly-Laminin is presented as a polymerized form of laminin, a protein associated with the development of the nervous system.
The Carlos Chagas Filho Foundation for Research Support of the State of Rio de Janeiro (Faperj) describes that laminin, most abundant in the embryonic phase, helps organize neural connections and becomes rare in the adult organism.
According to Faperj, the group’s strategy is to recreate, in the laboratory, a structure that functions as a support to guide the growth of nerve fibers after injury.
The foundation relates this proposal to the idea of fostering reconnection of circuits interrupted by trauma.
Public descriptions about the origin of the material converge on mentioning the placenta as a source of the protein.
According to publications related to the subject, laminin can be obtained from placentas, material that can be used for extraction and processing, and then transformed into poly-Laminin for application at the site of the injury in an experimental context.
A report from CNN Brasil recorded a statement from Tatiana Coelho Sampaio comparing the substance to other approaches under discussion, such as stem cell therapies, and mentioning aspects such as cost and standardization.

This comparison appears as a statement attributed to the researcher and, in the reports themselves, is presented in the context of a treatment still under investigation.
Another recurring point in the materials is the timing of the application.
Faperj states that the intervention was planned to occur close to the traumatic event, during surgery, and directly at the injured area, with rehabilitation following; the publication also indicates that the so-called “window” of time may influence the observed results.
Results in Humans: What Was Released About Tetraplegia and Movement Recovery
On social media, versions circulate claiming that six tetraplegics “started walking again.”
In institutional sources and in reports cited earlier, what appears with more consistency is that there were a small number of applications with recovery of movements to varying degrees, with emphasis on one case of returning to walking and reports of functional gains in other participants.
In a publication by Faperj, Bruno Drummond de Freitas reports that, weeks after the procedure, he was able to move his big toe and later started walking again.
In the same material, he attributes the recovery to the study: “I was the second person to receive this medication… it was thanks to this research… that I started walking again,” he said in an excerpt released by the foundation.
CAPES also reports that there were tests on people, in addition to experiments on animals, and emphasizes that the treatment is not available on a large scale and depends on new phases of study.
Meanwhile, CNN Brasil, when discussing the topic in September 2025, mentioned patients with different causes of trauma and described that the results released included partial or total mobility recovery in cases monitored in the experimental context, while pointing out the need for regulatory advancement.
Based on these publications, the claim that six people with tetraplegia started walking again does not appear confirmed with the same degree of detail and independent validation.
What can be found, documented in the consulted materials, is the description of motor improvements in a small group, with one widely reported case of returning to walking and other reports of recovery at varying levels.
Anvisa, Clinical Study, and Upcoming Tests with Spinal Injury
The regulatory advancement is presented as a necessary step to transform preliminary results into broader clinical evidence.
According to Adufrj, Anvisa authorized an initial phase aimed at assessing safety and potential side effects, with a limited number of participants, as is usual in the initial stages of clinical studies.
In the same communication, Tatiana Sampaio states that the goal is to expand research and investigate the effect on chronic injuries, which have been evolving for months or years.
This possibility appears as a research plan linked to the regulatory timeline and obtaining results in subsequent phases.
Reports also mention partnerships with health institutions for procedures and rehabilitation.
CNN Brasil cited the involvement of the Hospital das Clínicas of USP and AACD in predicted stages, in the context of the continuation of development and the structure needed to conduct studies.
Given the interest generated by the topic, the central question for researchers, patients, and health authorities becomes how the results will perform in larger studies, with standardized criteria and longer follow-up.

 Portuguese
Portuguese  English
English  Spanish
Spanish 





-

-

-

-

-

38 pessoas reagiram a isso.