Researchers from the University of Southern California created long-lasting progenitor cells of nephrons that grow stably in the laboratory and organize into three-dimensional renal organoids capable of filtering like a real kidney, opening the way to study genetic diseases, test drugs, and in the future, build artificial kidneys for the more than 94,000 Americans on the transplant waitlist
Researchers from the University of Southern California (USC) announced a breakthrough that could change the future of kidney disease treatment. They managed to create progenitor cells of nephrons that grow stably and for an extended period in the laboratory, something that has been one of the biggest obstacles in renal regenerative medicine so far. These cells are the starting material that builds nephrons, the filtration units of the kidneys, and the researchers used them to form organoids: mini three-dimensional structures that mimic the functioning of a real kidney.
The discovery does not mean that laboratory-grown kidneys ready for transplant are on the way tomorrow. But it means that researchers now have a more robust and scalable tool to cultivate kidney cells, study genetic diseases such as polycystic kidney disease, and test drugs with much greater precision than before. For the more than 94,000 Americans who were on the kidney transplant waiting list in December 2025, and for the millions of dialysis patients worldwide, any advancement in this direction is concrete. The study was published in the journal Cell Stem Cell.
What the USC researchers managed to do with kidney cells

Each human kidney contains about a million nephrons, microscopic structures that filter blood, remove waste, and balance water and minerals. When nephrons fail in sufficient numbers, the kidney stops functioning, and the patient needs dialysis or a transplant.
-
They were going to build a highway, but they ended up finding in the Czech Republic an entire Celtic city buried for more than 2,000 years, with hundreds of gold and silver coins, precious amber, and one of the largest archaeological treasures ever discovered.

-
Archaeologists find lost medieval city in Poland with a central square, cemetery, forge, and more than 500 buried artifacts.

-
A massive warm air mass stretched from Brazil to near Antarctica and caused something that almost never happens: lightning and thunderstorms in Ushuaia, in the Falkland Islands, and in the South Georgia Islands.

-
Larger than a football field, with a wingspan of 117 meters, six Boeing 747 engines, and a capacity of 250 tons, the Stratolaunch Roc turns the sky into a gigantic hypersonic launch ramp and accelerates the way the U.S. tests weapons from Mach 5 to Mach 10.

What the USC researchers did was cultivate the cells that build these nephrons so that they continued to grow and multiply in the laboratory for an extended period, without losing the ability to organize into functional structures.
To achieve this, the researchers adjusted the signaling of two proteins called p38 and YAP, which function as internal switches for the cells. By manipulating these switches, they kept the cells in a flexible and progenitor state, capable of continuing to divide.
The result was the long-term expansion of progenitor nephron cells from both mice and humans, including versions derived from human pluripotent stem cells that can be created from a simple blood or skin biopsy.
The renal organoids that the researchers created and why they are different
Renal organoids existed before this study, but most protocols generated a problem: along with the desired kidney cells, unwanted cells such as neurons and muscle fibers appeared.
The organoids created by the USC researchers showed only 0.67% off-target cells in single-cell analysis, a number drastically lower than previous protocols. This means purer mini structures that resemble a real kidney more closely.
The organoids also showed more pronounced maturation of podocytes, specialized cells that help filter blood within the nephron. And the researchers discovered something unexpected: the culture medium managed to induce mature podocytes to revert to a progenitor-like state.
This plasticity of kidney cells was something that researchers had not understood before, and it may open new possibilities for regenerating damaged kidney tissue.
How the researchers are already using the cells to test drugs
As the cells grew more and became easier to genetically edit, the researchers were able to use them for genomic screening via CRISPR. This technique allowed them to identify genes linked to kidney development and diseases, including already suspected genes and new candidates that had not been associated with the kidney before.
This is exactly the type of platform that researchers need when they want to understand how a kidney disease starts and what molecular targets can be attacked with drugs.
The researchers also modeled autosomal dominant polycystic kidney disease (ADPKD), the most common hereditary kidney disease, and evaluated 148 compounds for their effects on cyst formation.
Fourteen showed significant inhibition, 12 maintained efficacy in a second round, and PTC-209 stood out as an inhibitor that had not been previously associated with the suppression of ADPKD cysts.
For the researchers, this ability to test hundreds of compounds in pure and editable renal organoids is what transforms the discovery into a practical tool for the pharmaceutical industry.
What is still needed for researchers to build a complete artificial kidney
The researchers are clear about the limitations. The organoids are mini structures cultivated in the laboratory, not complete artificial kidneys ready for transplant.
There is still a need to generate the full range of nephron cell types, especially structures like the loops of Henle and the distal convoluted tubules, essential components for an artificial kidney to function like a real organ.
But the advancement is significant because it solves a problem that has stalled research for years: the difficulty of obtaining reliable kidney cells in sufficient quantities for study and genetic editing.
Zhongwei Li, the lead author of the study, stated that by enhancing the ability to cultivate these cells, researchers have created a new pathway to understand and combat congenital kidney diseases and cancer.
Chronic kidney disease affects more than 1 in 7 adults in the United States, and for these patients, every step toward an artificial kidney or more effective drugs is a step away from dialysis.
Cells that refuse to die and a future without dialysis
Researchers at USC created kidney cells that grow stably in the laboratory, organize into mini structures that mimic a real kidney, and are already being used to test drugs against hereditary kidney diseases.
It is not a ready-to-transplant artificial kidney, but it is the foundation on which this kidney can be built in the future, and for the millions of patients who depend on dialysis, the researchers’ message is that science is moving in the right direction.
Do you know someone who depends on dialysis? Do you think we will see artificial kidneys functioning in humans this decade? What do you think about the advancements made by USC researchers? Leave your thoughts in the comments and share this article with those interested in science and health.

 Portuguese
Portuguese  English
English  Spanish
Spanish 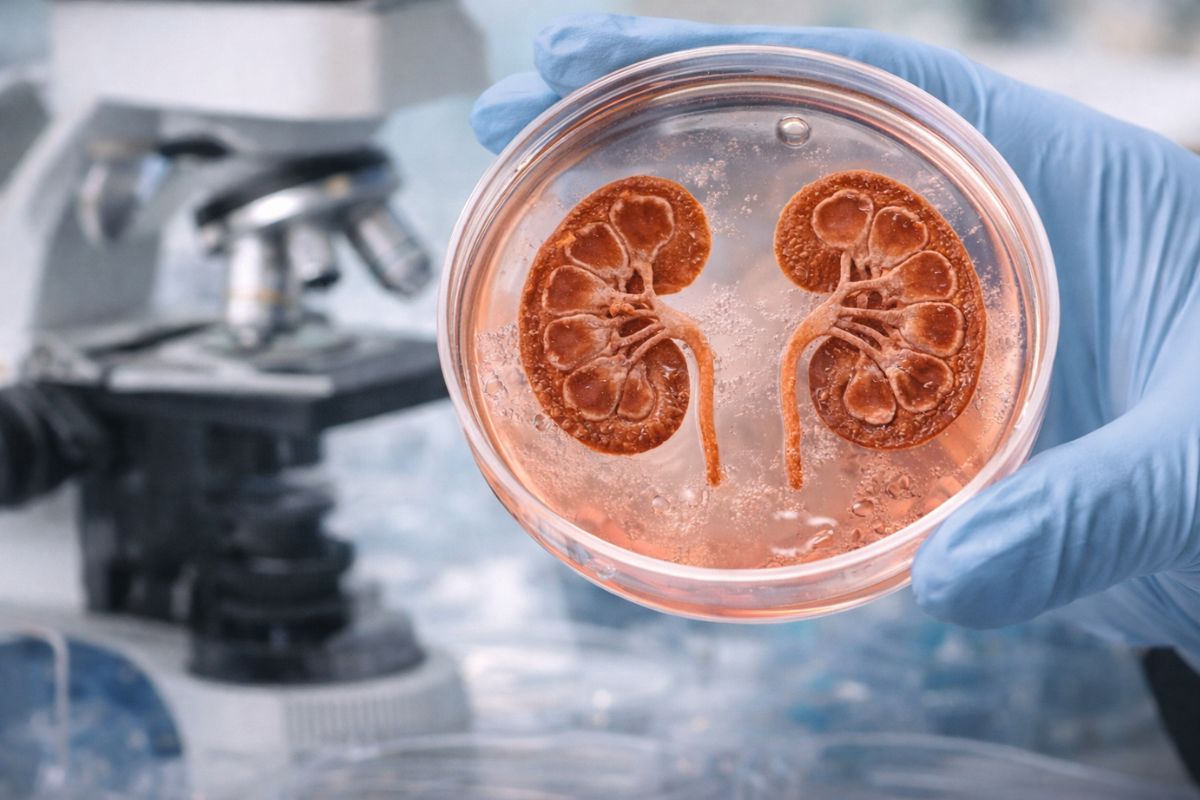




Seja o primeiro a reagir!