Scientific Study Details How the Action of a Specific Enzyme Triggers Genetic Chaos, Reorganizes Tumor DNA, and Hinders the Control of Certain Types of Cancer
A significant scientific discovery was announced in 2024 and has drawn the attention of the international medical community.
Researchers identified that the enzyme N4BP2 acts as a central factor in accelerating the evolution of certain cancers.
The study, published in the journal Science, helps explain why some tumors adapt quickly, escape therapies, and return in a more aggressive manner.
This finding reorganizes the understanding of the unpredictable behavior of cancer at the genetic level.
The research shows that the phenomenon occurs through a process known as chromothripsis.
In this mechanism, instead of slow and progressive mutations, an entire chromosome fragments abruptly.
Then, these fragments are reassembled chaotically, creating genetic shortcuts that favor tumor growth.
This type of event is rare in healthy cells but occurs frequently in cancerous cells.
Identification of the Genetic Trigger of Chromothripsis
For years, science understood the effects of chromothripsis but did not identify its direct cause.
This gap began to be filled with the investigation conducted by the University of California, San Diego, also reported in 2024.
The researchers analyzed, in real time, all the known human nucleases, enzymes capable of cutting DNA.
In this process, N4BP2 stood out unmistakably.
-
With a cost per shot close to zero, the DragonFire laser could change naval warfare in 2027 and provide British ships with nearly unlimited defense against drones.

-
A British startup creates tires that generate electricity in electric vehicles when passing over potholes, speed bumps, and cracks.

-
Scientists have created robots made with living cells that have their own nervous system, swim on their own, explore the environment, and self-organize without any genetic engineering, and now they want to do the same with human cells.
-
Students create a solar-powered ambulance that operates without a plug, without fuel, and still keeps medical equipment running in remote areas.

The enzyme demonstrated a unique ability to invade micronuclei, fragile structures that arise when chromosomes become isolated during cell division.
Without adequate protection, this exposed DNA becomes highly vulnerable.
In this environment, N4BP2 acts intensely, promoting rapid and successive cuts in genetic material.
This behavior explains the onset of chromosomal chaos observed in tumors.
Experimental Tests Confirm Causal Relationship
Experimental results reinforced the researchers’ hypothesis.
When N4BP2 was removed from brain cancer cells, chromosomal fragmentation significantly decreased.
On the other hand, when its presence was induced in healthy cells, intact chromosomes began to fragment.
In this way, it became clear that the phenomenon was not merely correlational but directly causal.
Moreover, tumors with high levels of the enzyme exhibited large amounts of extrachromosomal DNA (ecDNA).
These small rings of DNA carry genes that accelerate tumor growth.
They also contribute to resistance to drugs, hindering the success of conventional treatments.
According to the study, ecDNA may be a direct byproduct of chromothripsis.
Scientific and Clinical Impacts of the Discovery
In practice, the discovery shifts the focus of therapeutic strategies.
Instead of merely trying to contain established mutations, science starts to observe the initial moment of genetic disaster.
Blocking the action of N4BP2, or the pathways that allow its activity, emerges as a promising approach.
This strategy could reduce the tumors’ ability to adapt and make them more sensitive to current treatments.
Although it does not represent a definitive cure, the advancement offers a concrete possibility of cancer slowdown.
This slowdown could modify the clinical course of highly aggressive tumors.
According to the researchers, the discovery published in Science enhances the understanding of tumor evolution.
Thus, it opens up space for new strategies aimed at controlling cancer before it becomes practically uncontrollable.
In light of this scientific advancement, to what extent can controlling the enzymes responsible for genetic chaos redefine the future of aggressive cancer treatment?

 Portuguese
Portuguese  English
English  Spanish
Spanish 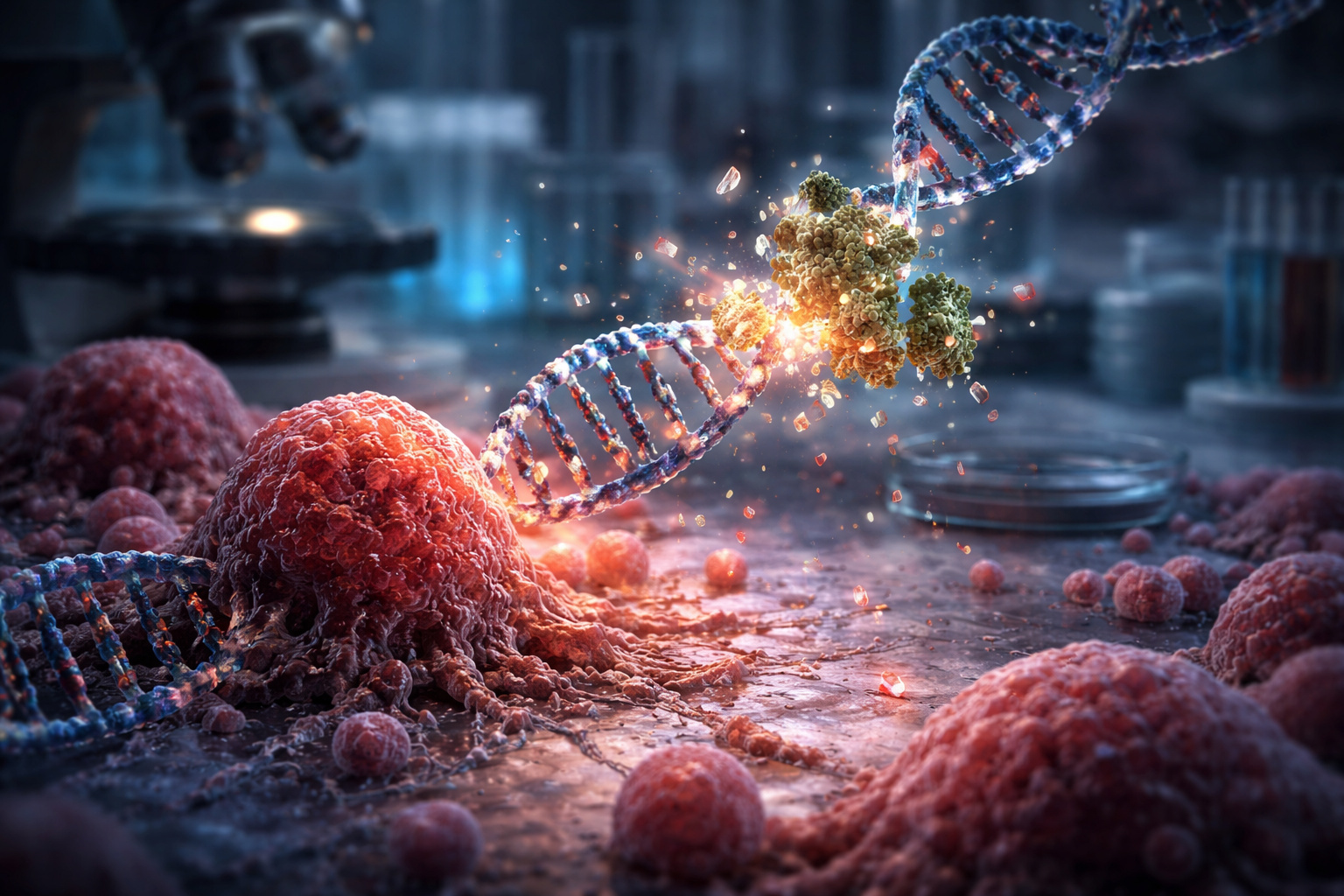





Seja o primeiro a reagir!