Study on cancer indicates that an inverted version of cysteine may act selectively on tumor cells, reduce tumor growth in mice, and open the way for a less aggressive complementary approach
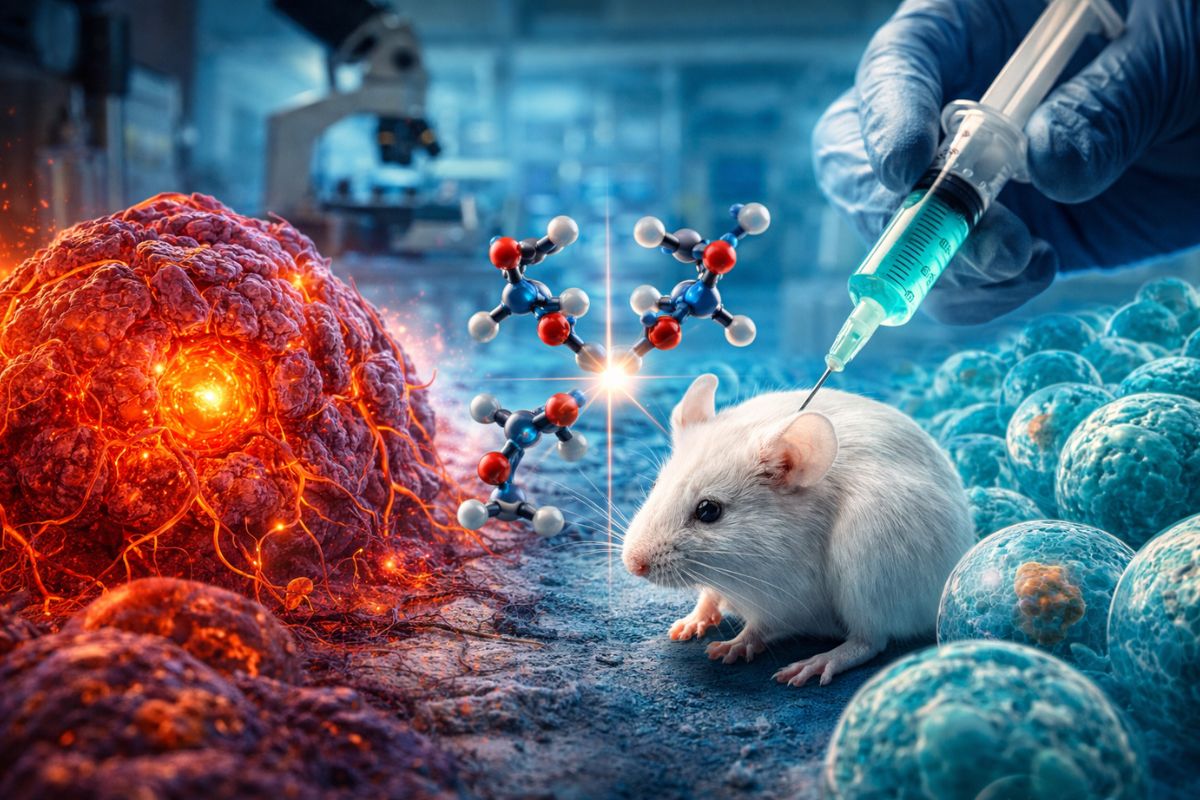
The cancer may gain a new front in the future from a strategy that deviates from traditional treatment patterns. Instead of indiscriminately attacking the body’s cells, the experimental proposal uses an inverted version of an amino acid to more precisely target tumor cells and preserve healthy tissues as much as possible.
The results are still preliminary, but they draw attention precisely because of this selectivity. In tests with mice, researchers observed a significant reduction in tumor growth without relevant signs of toxicity, which reinforces the potential of the technique as a possible complementary therapy in cancer cases.
How the new strategy against cancer works
The most well-known treatments for cancer often face the same challenge: to destroy the tumor, they frequently also end up affecting healthy cells. It is precisely at this point that the new approach attempts to differentiate itself.
-
350-year mystery may have been solved: remains of a soldier who inspired the hero of The Three Musketeers found beneath a church in the Netherlands.

-
NASA photographed a nearly perfect square with 3 km sides on Mars, 13 times larger than the Great Pyramid of Giza, but the official explanation is natural erosion; still, no one has explained why the four sides have almost identical lengths.

-
Starlink breaks the barrier of 10,000 satellites in orbit and takes its expansion to an unprecedented level, with a direct impact on global internet, technological competition, and the race for space.

-
Japan finds an alternative to oil amid rising prices by transforming ocean balance into electricity with a new technology that maintains efficiency even when the sea changes.

According to the study cited in the source, researchers investigated the potential of D-cysteine, a less common form of cysteine, to interfere with the metabolism of tumor cells.
The logic is to explore a vulnerability that seems to be more present in cancerous cells than in normal tissues, making the action more concentrated in the areas affected by the disease.
The role of the inverted version of the amino acid
The proposal relies on a well-known concept in molecular biology. Some substances can exist in mirrored versions, with the same chemical composition but different three-dimensional structures.
In the case of this experimental therapy for cancer, scientists observed that certain tumor cells have specific transporters capable of absorbing D-cysteine more easily.
This creates a more favorable entry point for the molecule precisely where the problem lies. This detail helps explain why the strategy may be more selective than other approaches.
What happens inside the tumor cell
After entering the cancerous cell, the molecule begins to act on the mitochondria, the structure responsible for energy production. The study points out that it inhibits the NFS1 enzyme, considered essential for cellular function.
In practice, this hinders the growth and division capacity of tumor cells. Instead of promoting immediate destruction, the strategy seems to act as a brake on disease progression.
This means that the observed effect against cancer is linked to slowing down the tumor, rather than necessarily to the direct elimination of cells.
Why selectivity draws so much attention
One of the main differentiators of this line of research is precisely the potential for selectivity. Since the entry of D-cysteine depends on a mechanism more present in tumor cells, the impact tends to concentrate in the affected regions.
This characteristic is relevant because one of the biggest obstacles in treating cancer is finding ways to act against the tumor without increasing damage to the rest of the body.
If this selective behavior is confirmed in future stages, the therapy could represent an important change in how to complement oncological treatment.
What the tests with mice showed
In the initial tests conducted with mice, researchers reported a significant reduction in cancer growth, without relevant signs of toxicity.
This is an important point because it combines two factors often observed in promising research: effect on the tumor and broader preservation of the organism.
Still, the results themselves require caution. The fact that the response was positive in animals does not mean that the therapy is ready for use in humans.
Between an experimental result and a clinical application, there is a long path of validation, safety, and efficacy.
Complementary therapy, not immediate replacement
Another important point is that D-cysteine does not seem to act as an isolated solution capable of replacing existing approaches against cancer.
According to the source, the most likely current use would be as a complementary therapy, in combination with already established treatments.
This possibility makes sense because the strategy acts by slowing down the growth of tumor cells. In a future scenario, this could expand the therapeutic arsenal available and help doctors and researchers think of more effective and less aggressive combinations.
The advancement, therefore, lies less in an immediate cure and more in opening a new avenue for intervention.
What this advance may represent
The research draws attention because it proposes a different way of dealing with cancer, targeting a metabolic characteristic of tumor cells and trying to exploit this difference in favor of treatment. It is a path that unites molecular biology, cellular metabolism, and the search for greater therapeutic precision.
Even in its early stage, the discovery reinforces an important direction in science: developing strategies that do not rely solely on brute force against the tumor, but on biological intelligence to better hit the target.
The more selective the therapy, the greater the chance of reducing collateral damage and preserving healthy tissues.
Do you believe that more selective therapies like this can decisively change cancer treatment in the future?






Seja o primeiro a reagir!