Revolutionary Discovery: Hydrogen Extracted Directly from the Ocean with Seawater Electrolysis and Wind Energy. Infinite Energy for a Sustainable World!
China can now produce hydrogen directly from the limitless oceans, harnessing energy from renewable sources like offshore wind energy.
Innovation in Seawater Electrolysis
Scientists from Shenzhen University and Dongfang Electric Innovation Institute (Fujian) achieved an incredible feat by developing a method called direct seawater electrolysis, which allows for hydrogen production directly from the vast oceans using renewable energy sources such as offshore wind energy.
Imagine the immense power and untapped potential that resides within our vast oceans, which cover over 96% of Earth’s water storage. With direct seawater electrolysis, we can tap into this colossal resource, extracting hydrogen – a clean and versatile fuel – directly from its depths. It’s like having an infinite energy supply waiting to be unleashed!
-
The Brazilian state accelerates industrial competitiveness with a focus on the extraction of strategic minerals to boost the energy transition in Goiás.

-
A Canadian retiree creates a hydroelectric system on a real river, generating energy continuously throughout the day and demonstrating how the power of water can supply a house with stability even in a simple structure.

-
Petrobras reaffirms its commitment to the market and ensures that it will carry out the energy transition safely to maintain national sovereignty.

-
Researchers discover a possible hydrogen deposit of up to 46 million tons beneath an ancient coal basin, and the volume could exceed half of the entire global production.

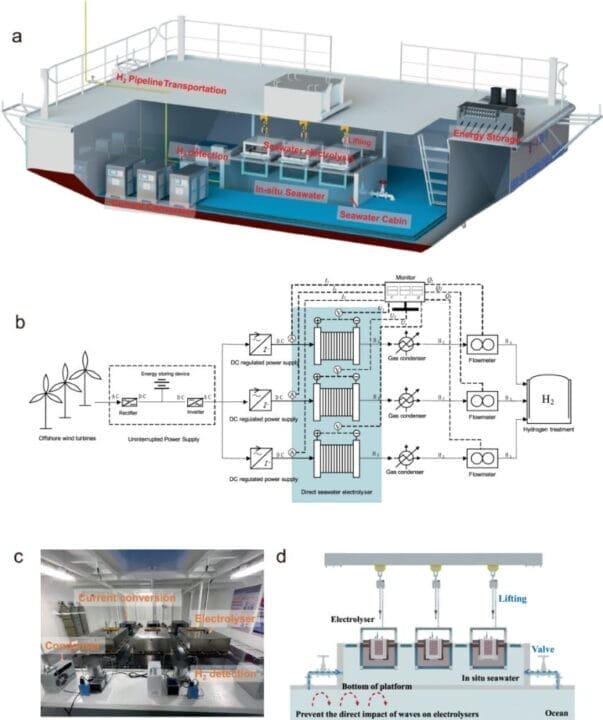
Integration with Offshore Wind Energy
But here is where the genius truly shines: scientists ingeniously integrated this technology with offshore wind energy. We all know that offshore wind farms have tremendous renewable energy potential, but effectively utilizing them has been a challenge. By combining the force of ocean waves with direct seawater electrolysis, we can now convert offshore wind energy into hydrogen energy on an impressive scale.
Direct Hydrogen Production
Direct hydrogen production from seawater using renewable energy sources like offshore wind energy has long been heralded as a potential solution for achieving a sustainable energy industry. While laboratory-scale experiments have shown promise, scaling the process and conducting in-situ demonstrations in the unpredictable ocean environment has presented significant challenges.
In this study published in Nature Communications, the team addressed the ocean’s floating conditions for the first time and successfully achieved seawater electrolysis in a wave-moving environment. They developed a floating platform equipped with a seawater electrolysis system powered by wind energy. The platform was deployed in Xinghua Bay, and a 1.2 Nm³/h pilot system was integrated for testing.
Impressive Results
Notably, stable electrolysis operation was achieved for over 240 hours under floating oceanic conditions, including wave heights ranging between 0 and 0.9 meters and wind speeds from 0 to 15 m/s. The system exhibited an electrolytic energy consumption of 5 kWh/Nm³ of H2 and produced high-purity hydrogen (>99.9%), comparable to land-based water electrolysis.
One of the main advantages of this advancement is the elimination of the need for pre-desalination processes. Traditional seawater electrolysis methods often require purification systems to remove impure ions, resulting in increased energy consumption and engineering costs. However, with direct seawater electrolysis, these additional steps are unnecessary, making in-situ hydrogen production in the oceans more feasible.
Overcoming Challenges
The researchers also tackled the complex composition of seawater, which contains numerous elements that can negatively affect electrolysis. Factors such as the presence of chloride ions and the deposition of insoluble substances during electrolysis can hinder hydrogen production efficiency. By overcoming these challenges, the team paved the way for more efficient and reliable direct seawater electrolysis.
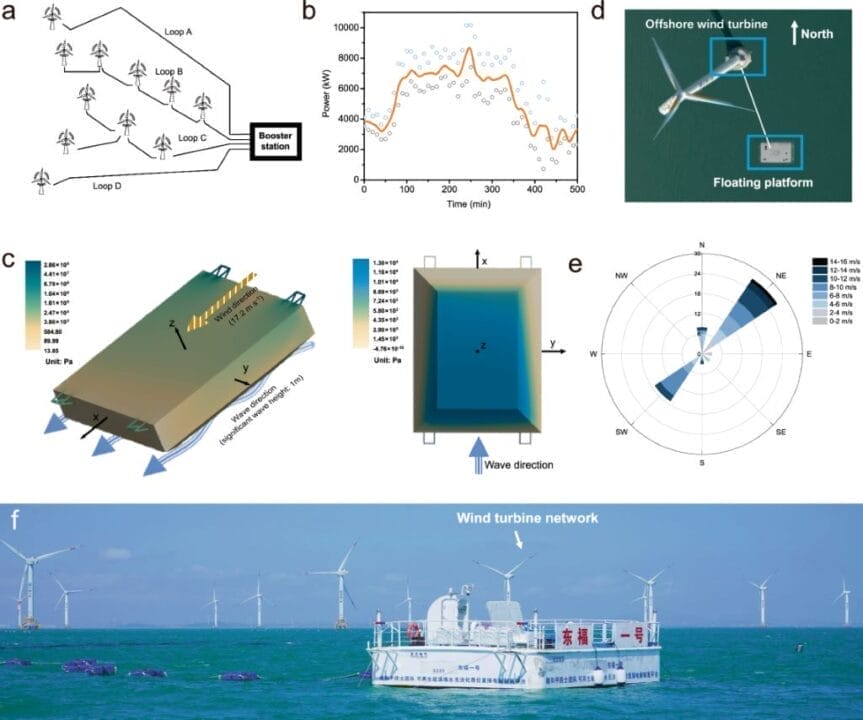
Toward a Sustainable Future
The successful demonstration of this floating seawater electrolysis system marks a significant step toward achieving a sustainable energy industry that harnesses the vast resources of the world’s oceans. It offers a promising alternative to fossil fuels and provides a pathway to convert offshore wind energy into clean hydrogen energy.
The researchers emphasized the importance of their findings in identifying technological challenges and the performance of key system components. They also highlighted the future prospects of this emerging technology, which could have far-reaching implications for the energy sector and contribute to global efforts to combat climate change.
This advancement in direct seawater electrolysis shows the potential to harness renewable energy sources and utilize the abundant resources of the oceans to achieve a greener, more sustainable future. Further research and development in this field are expected to enhance the efficiency and scalability of the system, bringing us closer to a world powered by clean, renewable hydrogen energy.
Source: www.nature.com
What Is Electrolysis?
Electrolysis is an electrochemical process that uses an electric current to induce a chemical reaction that would otherwise not occur spontaneously. This method is widely used in various industrial and scientific applications due to its ability to decompose chemical compounds and separate their basic components. The fundamentals of electrolysis and its main applications are explained below.
Fundamentals of Electrolysis
In basic terms, electrolysis involves using a direct current (DC) source to provoke a chemical reaction in a solution or melt that contains mobile ions. The electrolysis system typically consists of:
- Electrolyte: A substance that contains free ions and conducts electric current. It can be an aqueous solution of an ionic compound or a molten salt.
- Cathode: The negative electrode where positive ions (cations) are reduced by gaining electrons.
- Anode: The positive electrode where negative ions (anions) are oxidized by losing electrons.
- Electric Current Source: Provides the necessary energy to carry out the non-spontaneous reaction.
During the process, the ions from the electrolyte move toward the opposite electrodes under the influence of the electric current. At the cathode, the cations gain electrons (reduction), while at the anode, the anions lose electrons (oxidation).
Applications of Electrolysis
Electrolysis has numerous applications in various fields, including:
- Hydrogen Production: One of the most important uses of electrolysis is the production of hydrogen from water. This process is called water electrolysis and can be represented by the following overall reaction: [ 2H_2O(l) → 2H_2(g) + O_2(g) ]. The hydrogen produced is used as a clean fuel, as its combustion produces only water, without emitting pollutants.
- Metal Refining: In the metallurgical industry, electrolysis is used for refining metals such as copper, aluminum, and zinc. This process, known as electrorefining, enhances the purity of metals.
- Electroplating: Electroplating is the process of depositing a thin layer of metal onto the surface of another material using electrolysis. This is commonly used to improve corrosion resistance, appearance, and mechanical properties of metallic objects.
- Chlorination and Caustic Soda Production: The electrolysis of saline solutions, such as sodium chloride, produces chlorine and caustic soda (sodium hydroxide). This reaction is essential in the chemical industry for producing these important compounds.
Advantages and Challenges
Advantages
- Production of Pure Compounds: Electrolysis allows for the obtaining of high-purity elements and compounds.
- Efficiency and Control: The process is highly controllable and can be adjusted to optimize production.
- Versatility: It can be applied to a wide range of materials and compounds.
Challenges
- Energy Consumption: Electrolysis requires a significant amount of electrical energy, which can make some processes costly.
- Electrode Materials: Electrodes must be resistant to corrosion and other chemical effects, which can increase equipment costs.
Electrolysis is a crucial process in modern industry and science, with applications ranging from hydrogen production to metal refining and chemical manufacturing. Its ability to decompose compounds and produce pure elements makes it an invaluable tool in the quest for sustainable and efficient solutions.

 Portuguese
Portuguese  English
English  Spanish
Spanish 









Se der certo, isso abre grandes possibilidades. Com uma boa produção de hidrogênio seria possível converter as termoeletricas que usam carvão para usar o hidrogênio. Só na China já reduziria muito a geração de CO².