MIT Researchers Developed A New Battery Focused On Solar And Wind Energy Storage Systems That May Be More Economical And Efficient Than Conventional Batteries
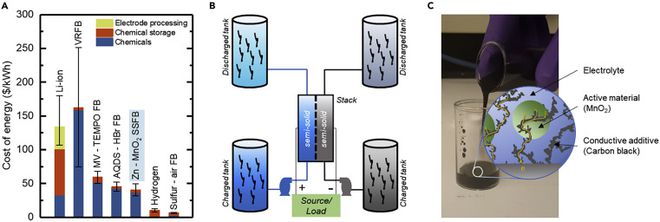
MIT researchers created a new type of battery that promises to innovate storage systems for clean energy sources such as solar and wind energy. The semi-solid flow cell uses a mixture containing dispersed manganese dioxide particles with an electrical conductive additive known as “carbon black.” Together, these compounds allow for the conversion of electrochemical electricity by reacting with other elements in aqueous suspension or zinc plates, enabling the creation of flow batteries with more effective, reliable, and long-lasting electricity retention systems than those manufactured today.
Also Read
Learn How The New MIT Solar And Wind Energy Battery Works
According to Emre Gencer, co-author of the MIT study, the transition to sustainable energies such as wind and solar necessitates batteries with different durations, that operate properly when the wind is not blowing and the sun is not shining.
-
Engineers propose a fourth traffic light for intersections – the change promises to affect crossings, queues, and waiting times.

-
Chevrolet Suburban Z71 lands in Brazil with a V8 engine, off-road design, air suspension, seven seats, and colossal size, a rare version imported for around R$ 1.3 million with luxury and exclusivity.

-
The Chevrolet Onix 1.0 turbo achieves 17.7 km per liter on the highway and ranks among the three most economical cars in Brazil in 2026, alongside two more expensive Toyota hybrids available at dealerships.

-
Little sister of the Hilux: Toyota launches Land Cruiser FJ with 4 radical versions, 2.7 engine with 166 hp and 4×4 traction with locking.

The rechargeable zinc-manganese battery is a cost-effective option for such sources, capable of storing energy for a day or more. In the flow system, two electrolytes with positive and negative ions are pumped in different tanks until they meet while passing through a membrane called the cell. The ion flows react with each other, transforming the electricity generated by solar or wind energy into chemical energy to charge the batteries.
The time it takes for the MIT batteries to discharge the stored energy depends on the volume of positive and negative electrolyte solutions flowing through the cell. As long as the movement is maintained, the batteries continue to supply electricity.
What Is Carbon Black?

According to Thaneer Narayanan, chemical engineer and lead author of the MIT study, for storage options lasting longer than a day, the architecture of flow batteries suggests they could be a more cost-effective option. There is the possibility to recharge the solution in the tanks using solar or wind energy sources, making the system carbon-free.
According to Thaneer, carbon black is a paste that adds pigment and electrical strength to the mixture. With this, the system is able to flow under reasonable pressures, maintaining a weak flow limit so that the active manganese particles do not settle in the flow tanks when the system is in use.
MIT Researchers Make Comparisons
To demonstrate that zinc-manganese batteries are economical, researchers compared them with other electrochemical cells and hydrogen-based storage systems. Analyses were conducted over durations of 8, 24, and 72 hours.
Over intervals longer than a day, the semi-solid flow battery surpassed the energy retention capacity of common vanadium redox flow cells and lithium-ion systems. It maintained low cost and efficiency, even considering all expenses related to pumping the manganese paste from the storage tank to the cell.










Seja o primeiro a reagir!