The Mini Livers Developed by MIT Scientists Have Already Shown, in Rat Studies, That They Can Remain Active for Eight Weeks, Produce Essential Substances, and Serve as Functional Support to a Compromised Liver, Paving the Way for a Less Invasive Alternative in Light of the Shortage of Compatible Organs.
The mini livers developed by MIT engineers put into perspective a possibility that until recently seemed far off: creating injectable grafts capable of taking over some functions of a compromised organ without immediately requiring a traditional surgical transplant. Instead of completely replacing the diseased liver, the proposal is to offer functional reinforcement to the body.
This idea gains traction because the liver performs an impressive volume of essential tasks for survival. When this organ loses efficiency, the impact spreads throughout the body. It is precisely at this point that the mini livers begin to attract attention, having demonstrated, at least in initial tests, the ability to produce important enzymes and proteins for consecutive weeks.
What Makes the Mini Livers So Relevant Right Now
The advancement draws attention because the liver is not a simple organ to support artificially. It is the largest solid internal organ in the human body and a structure responsible for about 500 fundamental functions related to the chemical regulation of blood. It participates in processes ranging from coagulation to the elimination of drugs and bacteria, which explains why liver failure is often so serious.
-
Scientists have finally discovered where the plastic that disappeared from the oceans went, and the answer is frightening: it fragmented into such tiny particles that they became invisible and now there are 27 million tons of nanoplastic just in the North Atlantic.

-
The Artemis II mission will take the Orion capsule back to the vicinity of the Moon with four astronauts, a private bathroom with a curtain, a system adapted for men and women, and a level of comfort that highlights how the Apollo era operated at the rawest limits of space exploration.

-
Giant structures on the seabed precisely control oil and reveal how high-tech underwater valves work.

-
China has just tested a giant drone the size of a fighter jet with a wingspan of 25 meters that carries 6 tons, flies for 12 hours, and the most frightening part: it can launch entire swarms of smaller drones directly from the air.

Much of this work depends on hepatocytes, specialized cells that perform central liver functions. Over the past decade, the team led by MIT researchers has been seeking a way to restore the activity of these cells without relying on complex surgery. The goal was not just to create tissue in the lab but to make that tissue survive and operate within the body.
The practical importance of this is evident given the number of patients waiting for a transplant. The material reports that over 10,000 Americans with chronic liver disease are in this queue amid a shortage of compatible organs. In this scenario, an intermediate solution capable of sustaining the patient until a donor arrives would already represent a significant clinical shift.
More than an abstract promise, researchers began to treat these structures as “satellite livers.” The expression helps to understand the logic of the project: instead of competing for space with the diseased organ or requiring its removal, the grafts would function as additional support, enhancing what the body can no longer do on its own.
How Scientists Managed to Transform Hepatocytes into Injectable Grafts
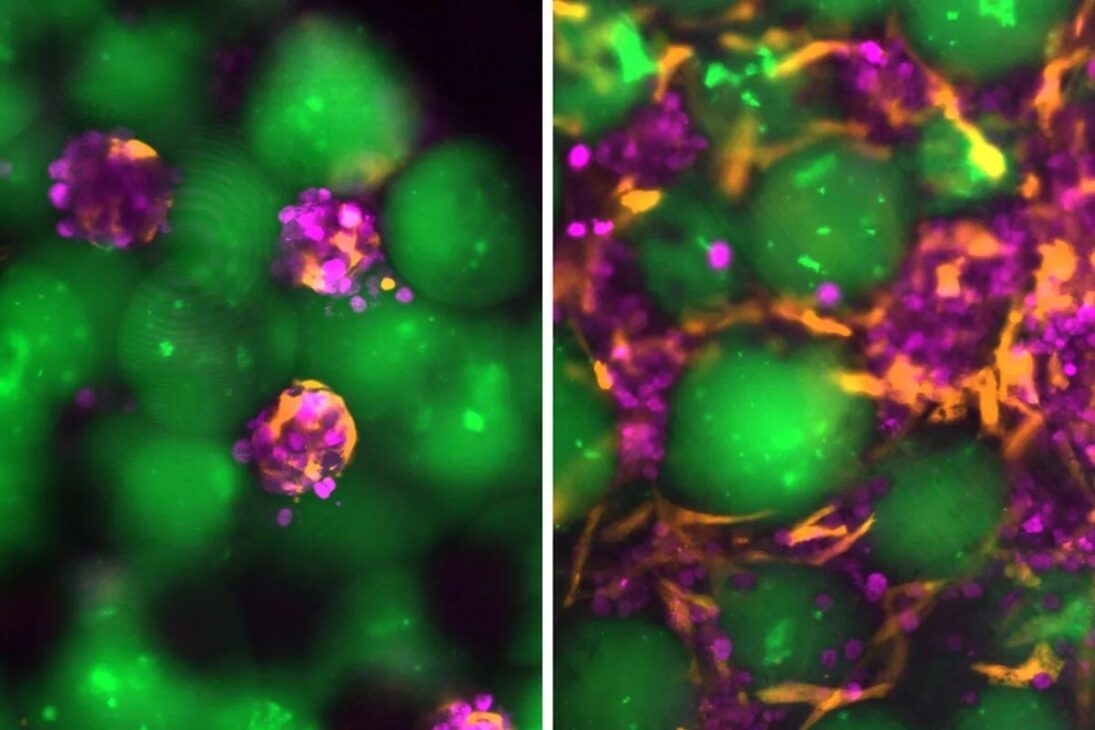
One of the main barriers was the very nature of cell transplantation. In previous experiments, one of the studied strategies was to incorporate hepatocytes into biocompatible materials such as hydrogels. These materials are three-dimensional networks of interconnected polymers that can absorb large volumes of water without dissolving, creating a favorable environment for the cells.
The problem is that, although useful for supporting hepatocytes, the hydrogels would require surgical implantation. This put the technique back into the realm of invasive surgery, precisely what the team wanted to avoid. The major breakthrough of the project was finding a way to inject not only the cells but the entire microenvironment necessary for their survival and integration into the organism.
The solution involved hydrogel microspheres. These structures have a particularly interesting behavior: when compacted, they behave like a liquid and can be applied with a syringe; after injection, they regain their solid structure. This property allowed the union of two needs that seemed difficult to reconcile: minimally invasive application and physical support for the transplanted cells.
In practice, these microspheres helped hepatocytes remain clustered, organized, and close to blood vessels. This is critical because isolated cells, released into the body without support, have much less chance of settling, receiving nutrients, and continuing to function. Without a niche, the cell does not integrate; with a niche, it gains a real chance of remaining alive and active.
The Role of Microspheres and Why They Make Such a Difference
The microspheres do not function merely as a transport vehicle. They create a favorable space for cells to migrate, settle, and form a stable graft. This behavior has already been considered promising even in wound healing, as it facilitates the occupation of spaces between the spheres by new cells and forming tissue.
In the case of the mini livers, this environment helped hepatocytes do something essential: quickly connect to the host’s circulation. This is central to any attempt at a functional implant because it is not enough to keep the cell intact. It needs to receive nutrients, exchange substances with the organism, and act in a biologically useful manner.
To further increase the chances of success, researchers added fibroblasts to the injectable mixture. These supportive cells helped improve the survival of hepatocytes and stimulated the growth of blood vessels in the graft. In other words, the team did not just inject liver cells; they assembled a small support system to help this tissue establish itself.
This detail helps to explain why the study advanced beyond a simple proof of concept. The new blood vessels formed alongside the hepatocytes, allowing them to receive nutrients directly, remain active, and produce the expected proteins. This is what distinguishes a cell present in the body from a cell that is effectively functional within the body.
Where the Grafts Were Placed and Why This Expands the Potential of the Technique
In experiments conducted with mice, researchers used the adipose tissue in the abdomen as the implantation site. The procedure was guided by ultrasound, both to position the syringe and to monitor the stability of the implant after application. This choice shows that the technique does not necessarily rely on the liver itself as the immediate destination for the cells.
This is one of the most relevant points of the proposal. According to the team, the grafts could also be implanted in other areas of the body, such as the spleen and areas near the kidneys. For most liver diseases, the graft does not need to be next to the liver to function, as long as it finds adequate space and has access to the bloodstream.
This anatomical flexibility increases clinical interest in the technology. Rather than being restricted to a single surgical window or a very specific point in the organism, therapy could be thought of in a more adaptable manner, considering the patient’s conditions and the feasibility of the procedure. This tends to reduce practical obstacles should the approach advance to future stages.
There is also an important shift in therapeutic logic. Instead of envisioning total and immediate repair of the damaged organ, the technique proposes a functional distribution: liver cells acting in another part of the body while still helping in metabolism and producing essential substances. It is a simple idea to understand and powerful in its implications.
What the Results in Rats Revealed After Eight Weeks
The strongest data presented so far is the observed activity time in the study. The injected hepatocyte cells remained operational throughout the analyzed period, which lasted eight weeks. During this time, the grafts successfully produced many of the enzymes and proteins normally made by real livers.
This matters because a test that is too short could indicate only transient survival. Here, however, the recorded behavior suggests functional continuity over a relevant period for an initial study. The graft not only remained in the organism but continued working. This distinction is crucial for assessing the potential of the technology as a long-term treatment.
This is still a result in animals, and this needs to be kept in perspective. Performance in rats does not guarantee the same response in humans, nor does it allow conclusions to be drawn that clinical application is near. But the findings lend concrete support to the hypothesis that injectable grafts may serve as a lasting aid to liver function.
It is also noteworthy that the proposal is not limited to cases of definitive replacement. The researchers themselves see the technology as a possible bridge to transplantation. This means providing temporary functional support until a compatible donor organ is available, which can be critical in situations of prolonged waiting.
Who Could Benefit Most If the Technique Advances to Clinical Use
The group most directly benefited would be patients with chronic liver disease who depend on transplantation and face a shortage of organs. For these individuals, any therapy capable of stabilizing body functions for longer may represent the difference between enduring until a donor arrives or deteriorating beforehand.
But there is another group that could gain ground in this discussion: patients who are not considered eligible for transplantation because they are too frail to tolerate surgery. In these cases, an injectable and less invasive approach could open an alternative where there are currently severe limitations. This point greatly expands the potential reach of the technology, as it is not just about reducing the waitlist but also including profiles previously excluded.
Researchers also highlight another advantage: if new grafts or complementary therapies are needed, the barrier to repeating the procedure tends to be lower than that of a new surgery. This changes the care strategy by transforming treatment into something possibly more adjustable over time.
Rather than a single radical intervention, what is emerging is a scalable approach. The patient could receive support, be re-evaluated, and, depending on the clinical response, undergo new applications with less impact than a conventional surgical intervention. This reasoning makes the mini livers not just a curious experiment but a therapeutic proposal with practical logic.
Current Limits and What Still Needs to Be Resolved
Despite the advancement, the technology still faces significant hurdles. In the current concept, patients would need to use immunosuppressive drugs to receive the mini livers. This point is important because immune rejection remains a central challenge in cell therapies and transplants.
In the future, the team is considering two possibilities to circumvent this problem. One would be to develop “stealthy” hepatocytes capable of evading immune rejection. The other would be to adapt the hydrogel microspheres to release immunosuppressants directly at the injection site, concentrating the effect where it is needed.
Both strategies show that researchers are not just looking at initial implant success but also at the clinical viability of the treatment in real-world scenarios. Still, there is a significant gap between good laboratory performance and large-scale adoption in human patients. This gap involves safety, efficacy, durability, and control of the immune response.
Therefore, the advancement needs to be viewed with moderate enthusiasm. There are objective reasons for attention and expectation, but there are also essential steps before this technology ceases to be an experimental result and begins to integrate into everyday medical care.
Why Mini Livers Generate So Much Expectation in Medicine
Mini livers attract interest because they combine, in a single project, three elements that rarely appear together with such balance: less invasive application, real biological function, and potential for use as prolonged support. It is not just about keeping cells alive in the lab but making them act as functional tissue within the organism.
This combination helps to explain why the topic seems like fiction at first glance. The idea of injecting small grafts capable of producing proteins and enzymes like a real organ sounds extraordinary. But what makes the story relevant is not the visual impact of the proposal, but its biological and clinical coherence. Mini livers were designed to solve a concrete problem: how to sustain patients when a transplant cannot happen immediately.
If the next steps confirm this potential, the technology could occupy a valuable space between conventional treatment and complete transplantation. This space is, today, precisely where many patients remain vulnerable, waiting for time, compatibility, and favorable clinical conditions.
From there, hepatic medicine could gain a new tool: not the immediate replacement of the organ but a functional reinforcement designed to save time, preserve stability, and expand therapeutic possibilities. And this, in itself, is already a transformative horizon.
The creation of mini livers by MIT shows how biomedical engineering has been trying to address one of the toughest challenges in hepatic medicine: providing real support to a weakened organism without exclusively depending on large-scale surgery.
The initial results in rats do not close this story, but they already change the way of seeing what may be possible in the coming years.
When a technology can unite biological precision, injectable application, and functional activity for weeks, it ceases to be just a scientific curiosity and deserves serious attention. Between the urgency of patients and the shortage of organs, mini livers emerge as a possibility that, for the first time, seems concrete.
Do you believe that injectable therapies like this can change the future of transplants, or is there still a long way to go before this becomes a reality?

 Portuguese
Portuguese  English
English  Spanish
Spanish 










Eu acredito sim, a medicina está muito avançada meus parabéns.