The proposal aims to expand the use of the brain implant in 2026, with mass manufacturing and a more automated implantation method
Elon Musk wants to take the brain chips from Neuralink to a new level starting in 2026, with large-scale production and an automated surgical procedure to implant the device.
The change could accelerate the availability of the implant for patients with severe mobility limitations, as it involves both mass manufacturing and automation of the implantation process.
What happened and why it got attention
Neuralink is one of the companies linked to Elon Musk, alongside Tesla, SpaceX, X Corp, and xAI, among others, totaling more than 5 companies in its portfolio.
-
Hummer H1X3: The 60-ton car, 14 meters long, and $25,000 tires – all for a single owner in the United Arab Emirates.

-
A city in the United States has a secret to handle peak electricity demand and can supply power to about 300,000 homes for up to 4 hours.

-
In the desert, at an altitude of 5,000 meters, there is a radio telescope that consumes as much energy as a city and relies on artificial oxygen to operate.

-
Ancient remains found 200 meters in a cenote chamber are retrieved from the sea and reveal a human skeleton in a shelter or ritual space from 8,000 to 10,000 years ago.

Now, the proposal is to initiate a phase of mass production of the brain chips in 2026 and advance to an automated surgery model.
The topic draws attention because it involves implants in the brain with a direct interface between mind and computer, something that for a long time seemed distant from real use.
How Neuralink got here
The attempt to implant the chip in humans faced regulatory barriers for years, with the FDA blocking progress to human trials at different times.
During this period, tests were conducted on animals, including demonstrations where monkeys managed to play Pong using their minds.
The release for human use occurred, and in 2024, the first human with a Neuralink chip was seen, although problems arose after this milestone.
How the brain-machine interface works in practice

The central idea is to connect the brain to an interface that allows communication and control of devices using neural signals.
In practice, this paves the way for tasks such as operating a computer and, with training, even playing video games, maintaining activities that many people believed would be lost after paralysis.
The focus is a very specific use, aimed at those with limited mobility, and not a widely consumed product.
What changes in practice for patients with severe paralysis
Neuralink operates in a niche, but demand has reportedly grown to the point of motivating the transition to large-scale production.
In addition to manufacturing, the plan includes an automated procedure for implanting the chip, indicating an attempt to standardize and accelerate the surgical stage.
The practical promise of this model is to reduce bottlenecks in implantation, as the process no longer depends solely on a traditional surgical flow.
What are the rules, deadlines, and conditions mentioned
The central milestone mentioned is 2026, the year expected to begin large-scale manufacturing and the adoption of the automated implantation method.
There is also a current adoption figure: there are 12 patients in the world using the brain chips from Neuralink, all with severe paralysis.
In cases of chip failures in one of the patients, it was reported that there was a correction through software updates, which keeps the issue of reliability in evidence.
What could happen from now on
The proposed scale tends to put more pressure on safety, stability, and long-term monitoring, as future results are still unknown.
The plan gains more context after the company raised 650 million dollars in a funding round in June 2025, reinforcing the capacity to finance expansion.
If mass production and automated implantation proceed as planned, Neuralink could expand the reach of the implant within the audience it already serves today.
The central point is that Elon Musk aims for 2026 to scale Neuralink, combining mass manufacturing and automated surgery in the implantation of the chip.
With 12 patients already implanted and 650 million dollars in funding in June 2025, the company enters a phase where the impact could grow, but still with natural doubts about long-term results.

 Portuguese
Portuguese  English
English  Spanish
Spanish 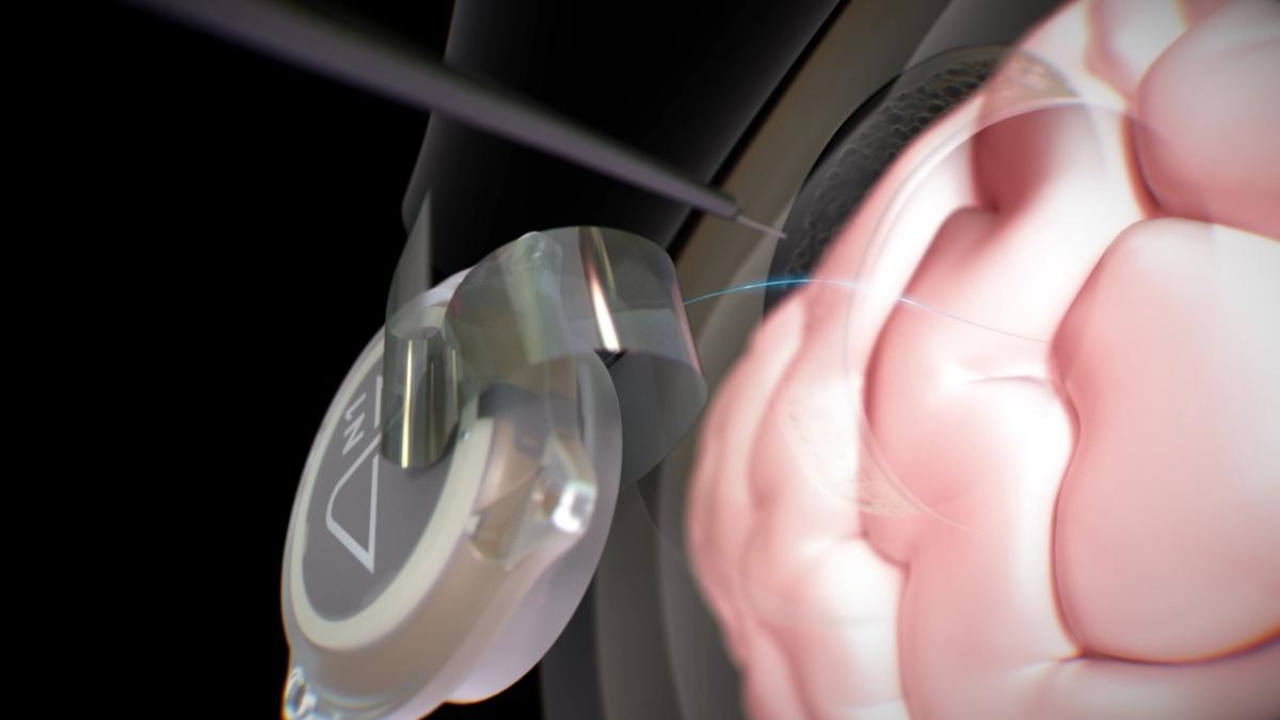






Seja o primeiro a reagir!