Ancient Malice, Modern Cure: Egyptian Tomb Fungus May Be the Next Revolution Against Cancer
Researchers have discovered that the same fungus associated with the so-called “curse of the mummies,” responsible for mysterious deaths of scientists last century, contains compounds with high potential for treating blood cancer. The villain of archaeology may now turn into a hope in medicine.
A study published in Nature Chemical Biology revealed that Aspergillus flavus, a fungus known for producing deadly toxins, has molecular structures capable of destroying leukemia cells without significantly affecting other types of tissue. This finding paves the way for a new class of drugs that is still in its early stages but holds real potential for oncological treatment.
The Dark Legacy of Tutankhamun’s Tomb
The origin of fear surrounding Aspergillus flavus dates back to the 1920s during the famous expedition that uncovered the tomb of Pharaoh Tutankhamun in Egypt. After the opening of the site, several crew members began to die under strange circumstances, giving rise to the theory of the “curse of the mummies.”
-
Brazilian city neighboring Paraguay becomes a ‘magnet’ for people seeking quality of life and high standards with exceptional growth and commutes of up to 15 minutes.

-
A rare storm transformed part of the Gulf into an unlikely scene of hail, extreme rain, and tornado risk in the middle of the desert, with volumes that exceeded the annual average in a single day.

-
Brazil catches the world’s attention with ships powered by ethanol and biodiesel that have lower emissions, an unprecedented certification with advantages in chartering, and a revolution in cabotage.

-
A couple who only has Sundays free has built a hand-dug pool, a hamburger joint, a dance hall, and a playground in their own home without hiring a mason, and even with breaks due to accidents and the pandemic, the work has never truly stopped.

Decades later, a similar event occurred in Poland: ten scientists died after exploring the tomb of King Casimir IV. Later analyses found high concentrations of Aspergillus flavus spores in the enclosed environments of these tombs. The fungus releases toxins that attack the respiratory system, potentially leading to death in individuals with weakened health.
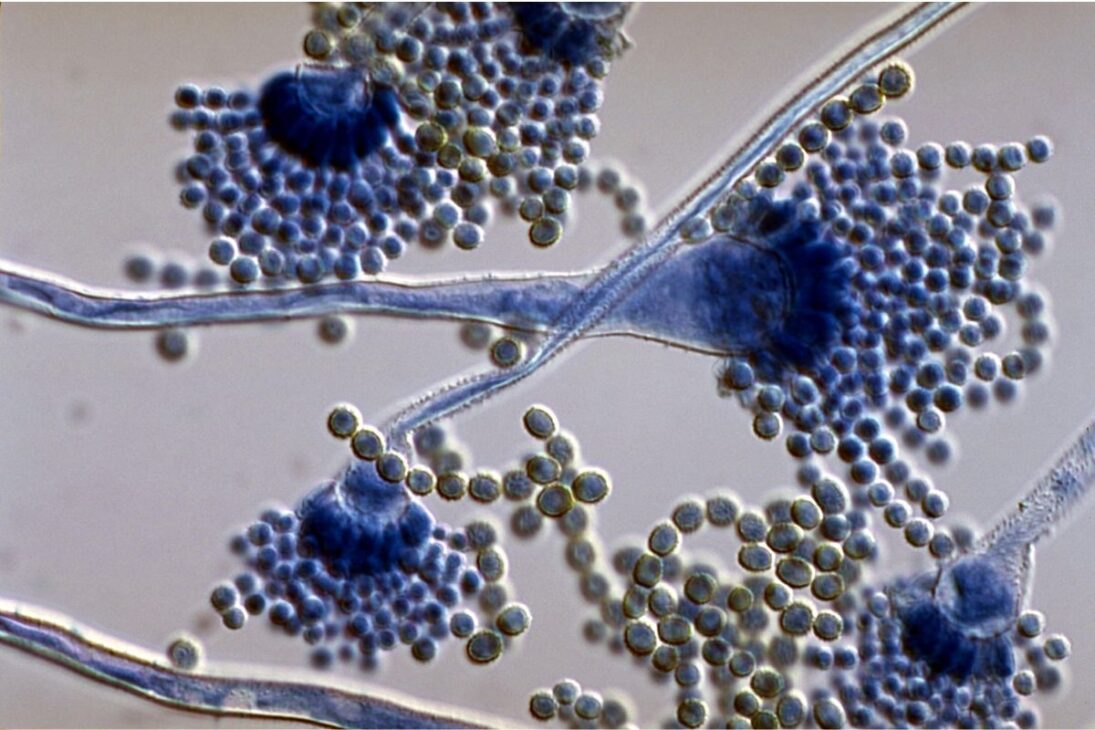
From Archaeological Threat to Therapeutic Molecule
The fact that toxic substances can be converted into medicines is not new in science. Penicillin itself, extracted from Penicillium, is also derived from a fungus. The difference now lies in the complexity of the newly discovered molecule, named aspergillomarine.
This is a class of compounds called ribosomally synthesized and post-translationally modified peptides (RiPPs). These microcompounds act as “mini-proteins” with a unique architecture of intertwined rings. Researchers successfully isolated four variants of these RiPPs and, by modifying them with lipids, created an effective structure against leukemia cells.
According to the scientists involved, aspergillomarine interferes with the process of cell division by blocking the formation of microtubules — essential structures for separating chromosomes during mitosis. The result is programmed cell death, a fundamental mechanism for curbing the advancement of leukemia.
Comparable Potency to Approved Medications
The new compound showed, in laboratory tests, effectiveness similar to that of established drugs in leukemia treatment, such as cytarabine and daunorubicin, both approved by the FDA. However, aspergillomarine’s action appears more selective: it showed efficacy almost exclusively against leukemia cells, with little or no action against breast, liver, or lung cancer cells.
“It’s very encouraging to find a cytotoxic agent with this degree of specificity,” said hematologist José Larios from the Barbara Ann Karmanos Cancer Institute in the U.S. Despite the optimism, the expert reminds that the pathway to the approval of a new drug is long and rigorous.
Producing on a Large Scale Is Still a Challenge
Besides the scientific steps, there is a practical barrier: large-scale production. According to Larry Norton, Vice President of the Memorial Sloan Kettering Cancer Center, drugs derived from fungi have a tradition in medicine — penicillins, cephalosporins, and statins are good examples. However, when it comes to compounds as specific as aspergillomarines, cultivating the fungus and extracting the active principle may become financially unfeasible.
The solution, says Norton, would be to develop methods of artificial synthesis of the compounds, a chemical engineering endeavor that is still in early stages but is essential for the drug to become commercially viable.
A 10-Year Journey to Hospitals
Following traditional protocols, the substance will now need to undergo pre-clinical tests, including cell cultures and animal models, to assess toxicity, dosage, and efficacy. Only then can it advance to clinical trials in humans — which include phases I, II, and III — and, if all goes well, obtain FDA approval for commercial use.
Data from the Journal of the National Cancer Institute show that only 10% of drugs that reach phase II trials succeed in obtaining final approval. Nevertheless, the current context pressures science to seek alternatives: many cancers become resistant to available treatments, demanding constant innovation.
“Even when we manage to control certain types of cancer with initial treatment, in many cases, the body finds ways to evade the drugs. Therefore, new therapeutic approaches are always welcome and necessary,” concluded Larios.

 Portuguese
Portuguese  English
English  Spanish
Spanish 








-

-

-

-

-

5 pessoas reagiram a isso.