Developed in Brazil, Polylaminin Enters Clinical Trials and Emerges as Hope in the Treatment of Paralysis Caused by Spinal Cord Injury.
The polylaminin, a molecule developed by Brazilian researchers, has officially entered phase 1 of clinical trials after approval from Anvisa, representing a new hope for people with spinal cord injury and paralysis.
The drug, created from a natural protein in the human body, began human trials this year in Brazil after nearly three decades of scientific research conducted by the Federal University of Rio de Janeiro.
The aim is to rigorously evaluate whether the substance is safe and may, in the future, help in the recovery of lost movements after severe spinal cord trauma.
-
With a cost per shot close to zero, the DragonFire laser could change naval warfare in 2027 and provide British ships with nearly unlimited defense against drones.

-
A British startup creates tires that generate electricity in electric vehicles when passing over potholes, speed bumps, and cracks.

-
Scientists have created robots made with living cells that have their own nervous system, swim on their own, explore the environment, and self-organize without any genetic engineering, and now they want to do the same with human cells.
-
Students create a solar-powered ambulance that operates without a plug, without fuel, and still keeps medical equipment running in remote areas.

Polylaminin and the Challenge of Spinal Cord Injury in Brazil
Spinal cord injury is one of the most disabling conditions in medicine.
In general, it occurs after traffic accidents, falls, diving into shallow water, or episodes of violence, such as gunshot wounds.
Depending on the severity and the affected region, the condition may progress to partial or total paralysis, including paraplegia and tetraplegia.
Currently, there is no treatment capable of fully restoring movement in cases of complete spinal cord injury.
Surgery and rehabilitation help preserve functions and quality of life, but do not promote nerve regeneration. It is precisely in this scenario that polylaminin draws the attention of the medical community.
What Is Polylaminin and How Does It Work
Polylaminin is a compound derived from laminin, a protein naturally produced by the human body, especially during embryo formation.
According to researcher Tatiana Sampaio, the study leader, this protein is essential for the growth of axons — structures responsible for transmitting signals between neurons.
“You have a protein that is already very powerful, and we have it now in a, let’s say, improved form in the lab,” explains the scientist.
The idea is for polylaminin to function as a “biological scaffold,” creating a favorable environment for axons to regrow after injury.
Brazilian Scientific Research and Initial Results
Before reaching clinical trials, polylaminin underwent years of laboratory experiments and testing on animal models.
Consistently, the results indicated axon regeneration, regardless of the type of spinal trauma analyzed.
“It always worked. Regardless of the type of injury, the axons grew back. That’s what gave us confidence to move forward,” said Sampaio.
In a preliminary study with eight human patients, conducted by UFRJ in partnership with the pharmaceutical company Cristália, six showed recovery of movements.
The response rate of 75% far exceeded historical data, which indicate spontaneous improvement in about 15% of similar cases.
Judicial Liminaries and Cases That Gained Attention
Even before commercial approval, some patients gained access to polylaminin through court decisions.
One of the best-known cases is that of nutritionist Flávia Bueno, who regained movement in her right arm after receiving the application of the substance at Albert Einstein Hospital in São Paulo.
Despite positive reports, Sampaio issues a warning.
To her, the use outside of formal protocols is “wrong, inappropriate, and improper” from a scientific standpoint, as it may compromise data collection and the assessment of adverse effects.
How the Clinical Trials of Polylaminin Work
Phase 1 of the clinical trials will evaluate the safety of the treatment in five patients with complete thoracic spinal cord injury. The application occurs during emergency surgery, up to 72 hours after the trauma.
The studies are supported by the Hospital das Clínicas of the Faculty of Medicine of USP, Santa Casa de São Paulo, and AACD, responsible for the rehabilitation of the patients.
If the results are positive, the research will advance to phases 2 and 3, which assess efficacy and safety in a larger number of people.
Expectation, Caution, and Next Steps
The researchers expect to conclude phase 1 by the end of this year. If everything goes as planned, the registration request may be submitted to Anvisa in 2028.
For experts not directly involved in the study, such as physiatrists Ana Rita Donati, the moment requires caution.
She notes that some improvement can occur even without new medications and that definitive conclusions require time and more patients evaluated.
Still, polylaminin represents one of the most promising advances in scientific research in Brazil in the neurological field.
As the studies continue, the expectation grows among patients, doctors, and researchers who see, for the first time, a concrete possibility of changing the treatment of paralysis caused by spinal cord injury.

 Portuguese
Portuguese  English
English  Spanish
Spanish 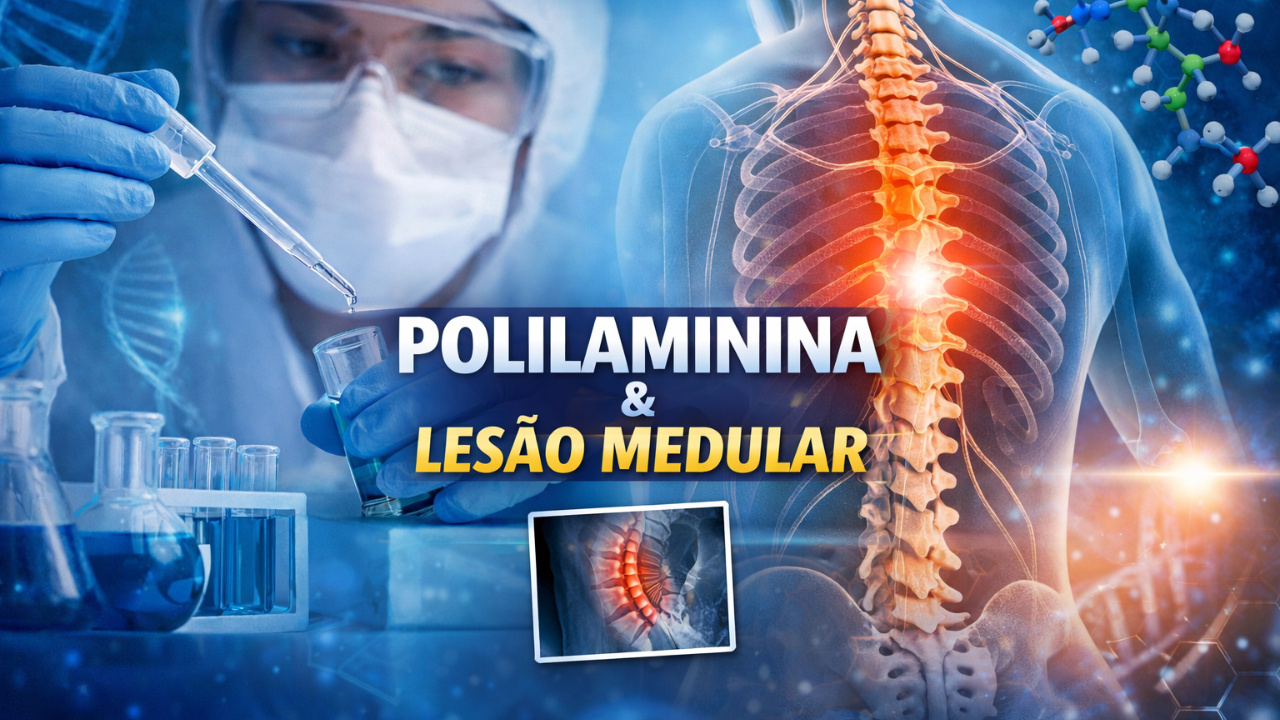






-

-

2 pessoas reagiram a isso.