By reproducing the real porosity of bone in structures of 500 nanometers, a hydrogel created in Switzerland shows that lighter, more precise, and temporary implants could change clinical recovery in the coming years.
A team from ETH Zurich has developed a hydrogel capable of mimicking how bone heals in the body. The proposal paves the way for implants that are better tailored to the patient and have a more natural response in the organism.
The material combines 97% water and a small fraction of biocompatible polymers. When exposed to laser pulses, it gains enough structure to serve as a base for bone regeneration in more complex cases.
In practice, this can reduce extra steps in treatment and improve the adaptation of the implant to the patient’s own bone cells. The advancement also helps shorten the time between manufacturing and clinical application.
-
A gigantic freshwater reservoir appears beneath the Great Salt Lake, between 3 and 4 kilometers deep, surprising scientists with an unexpected flow towards the interior of the lake.

-
At over 8,800 meters above sea level, the summit of Mount Everest is made up of rocks that originated at the bottom of an ocean about 500 million years ago and were pushed to the highest point on Earth by the collision of tectonic plates.

-
Japan targets a remote island in the Pacific measuring only 1.51 square kilometers, located 1,900 kilometers from Tokyo, to store nuclear waste for up to 100,000 years, raising alarms at sea.

-
A 1,900-year-old papyrus found in the Judean Desert reveals a crime of the Roman Empire involving fraud, forgery, and a detailed court hearing.

Tiny structures mimic bone architecture
The researchers managed to produce structures of 500 nanometers, a scale that approaches very fine elements of bone tissue. This detail makes a difference because it increases the chance of cells adhering to the material more efficiently.
With this support, bone cells begin to occupy the space and produce collagen, an essential component for recovery. The hydrogel acts as a temporary base that follows the natural logic of healing.
Material dissolves after fulfilling its function

Another important point is that the implant was not designed as a permanent piece. After assisting in regeneration, the hydrogel tends to gradually dissolve within the body.
This behavior favors a more organic repair and decreases dependence on rigid structures. For the patient, the main impact is a recovery with greater biological integration and less artificial interference.
Printing speed changes the rhythm of implants
The team achieved 400 mm per second in the manufacturing of biological structures, a number that stands out for the scale gain without losing precision. This allows for the production of very fine geometries in less time.
According to Advanced Materials, an international scientific journal focused on materials research, the development describes a system capable of creating implants at high speed with extremely high resolution. This performance strengthens the use of customized parts in orthopedics.
Fine precision helps cells occupy the implant
The technology also works with very small resolution, finer than the diameter of a human hair. This level of detail is crucial for reproducing the porosity that exists in natural bone.
When the structure better mimics the channels of bone tissue, the organism’s response tends to be more favorable. The expected result is faster regeneration with greater cellular adherence from the very first stages.
Initial tests show integration with bone cells
In laboratory trials, the samples indicated that the material is biocompatible and can receive bone-forming cells without compromising the created structure. This reinforces the potential of the hydrogel as a base for new implants.
The results still belong to an initial phase, but already support a next, more demanding stage. The proposal now is to verify if the same behavior appears under more complex biological conditions.
Next step involves animal testing
The research advances in partnership with the AO Research Institute in Davos to observe cellular migration and the recovery of skeletal strength in living organisms. This phase is crucial for measuring performance outside the laboratory.
If the data is confirmed, the use of customized bone implants could gain speed and safety in future treatments. This repositions regenerative medicine and changes the strategic outlook.

 Português
Português  Inglês
Inglês  Espanhol
Espanhol 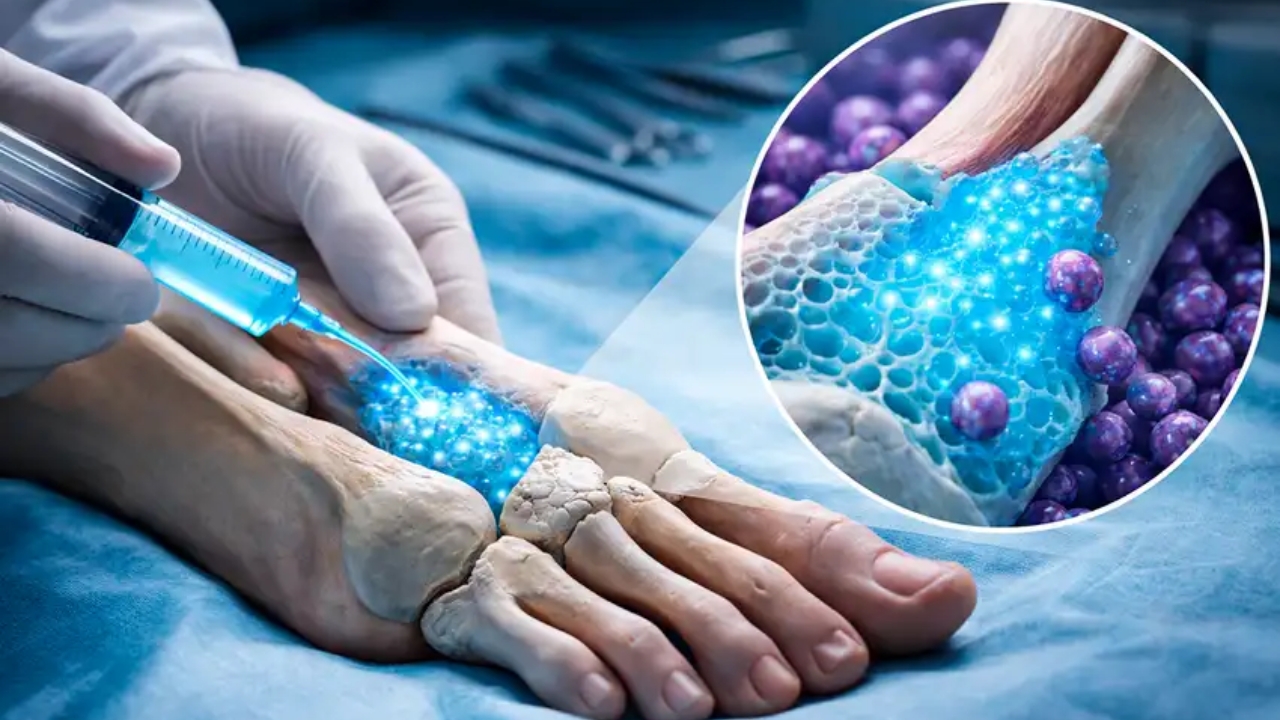





Seja o primeiro a reagir!