Anvisa Prohibited 32 Supplements from Ervas Brasillis Due to Unsanitary Conditions and Lack of License. Know Which Brands Were Affected and the Health Risks.
The National Health Surveillance Agency (Anvisa) determined, last Friday (09/05/2025), the prohibition of the manufacturing, sale, import, and use of 32 dietary supplements produced by Ervas Brasillis Produtos Naturais Ltda.
The measure, published in the Official Gazette of the Union, applies to all batches and includes the immediate seizure of products in circulation.
The decision was made after a health inspection revealed that the supplements were made without a health license and under unsanitary conditions, violating the good manufacturing practices required by Brazilian legislation.
-
Cosulati’s plant gains new life with a million-dollar investment and a plan to process up to 550,000 liters of milk per day.
-
Could chocolate disappear from the world? Understand why cocoa could vanish in the coming decades and transform the sweet into a rare item.
-
Chinese companies are sharing factories in Brazil to avoid import taxes, reduce costs, and use the country as a production base to sell cheaper in the domestic market.
-
Usiminas closed a contract to supply 5,200 tons of special steel for the four most advanced frigates the Brazilian Navy has ever built — each ship carries 1,300 tons of plates that need to resist the sea and explosions.
Why Did Anvisa Suspend the Supplements?
According to Anvisa, inspectors found serious irregularities during the inspection. The supplements were produced in a location without health authorization, which characterizes illegal activity.
In addition, the lack of hygiene and quality control posed risks to consumer health.
The agency emphasized that, in addition to the suspension, any form of advertising or distribution of these products is prohibited.
This means that pharmacies, supplement stores, e-commerce sites, and resellers must immediately remove the items from the market.
What Does the Legislation Say About Health Surveillance?
According to Article 46 of Decree-Law 986/1969, any factory or company that manipulates or transports food needs to have prior authorization issued by municipal, state, or district health surveillance.
The lack of this authorization makes production illegal and subject to penalties such as seizure and interdiction.
This rule exists precisely to protect consumers from risks associated with the manufacturing of food and supplements in environments without adequate conditions.
Supplement Brands Affected by the Decision
The list of products suspended by Anvisa is extensive and involves different brands linked to Ervas Brasillis.
Among them are Turbo Black Vitamin, NB Nutrition, Ervas Brasil, Natuforme, Ozonlife, Nutritional Sports, Max Force, and Vitacorpus.
Check out some of the main prohibited supplements:
Creatine Monohydrate (red fruits and orange flavors) – Brand Turbo Black Vitamin;
Undenatured Type II Collagen – Brands Turbo Black and NB Nutrition;
Caffeine with Taurine – Brand Turbo Black Vitamin;
Peruvian Maca, Black Maca, and Premium Maca – Brands Ervas Brasil, Turbo Black, Nutritional Sports, and Max Force;
Hibiscus and Graviola – Brand Ervas Brasil;
Magnesium Threonate and Magnesium Dimaleate – Brands Turbo Black and NB Nutrition;
Omega 3 – Brand NB Nutrition;
Ora-Pro-Nobis Premium – Brand Vitacorpus;
Ozonized Sunflower Oil – Brand Ozonlife.
All batches of these formulas are prohibited from circulating in the market.
Risks to the Consumer
The use of supplements manufactured under unsanitary conditions can pose serious health risks.
The lack of quality control can result in contamination, incorrect nutrient dosages, or even the presence of undeclared substances on labels.
For health surveillance specialists, this situation highlights the importance of choosing supplement brands with registration and authorization from Anvisa, ensuring that they have undergone quality and safety testing.
What Should Consumers Do Now?
Anvisa’s recommendation is clear: anyone who purchased any supplements from the prohibited list should immediately stop consuming them.
Additionally, consumers can return to the place of purchase for a refund or report the illegal sale to health authorities.

 Portuguese
Portuguese  English
English  Spanish
Spanish 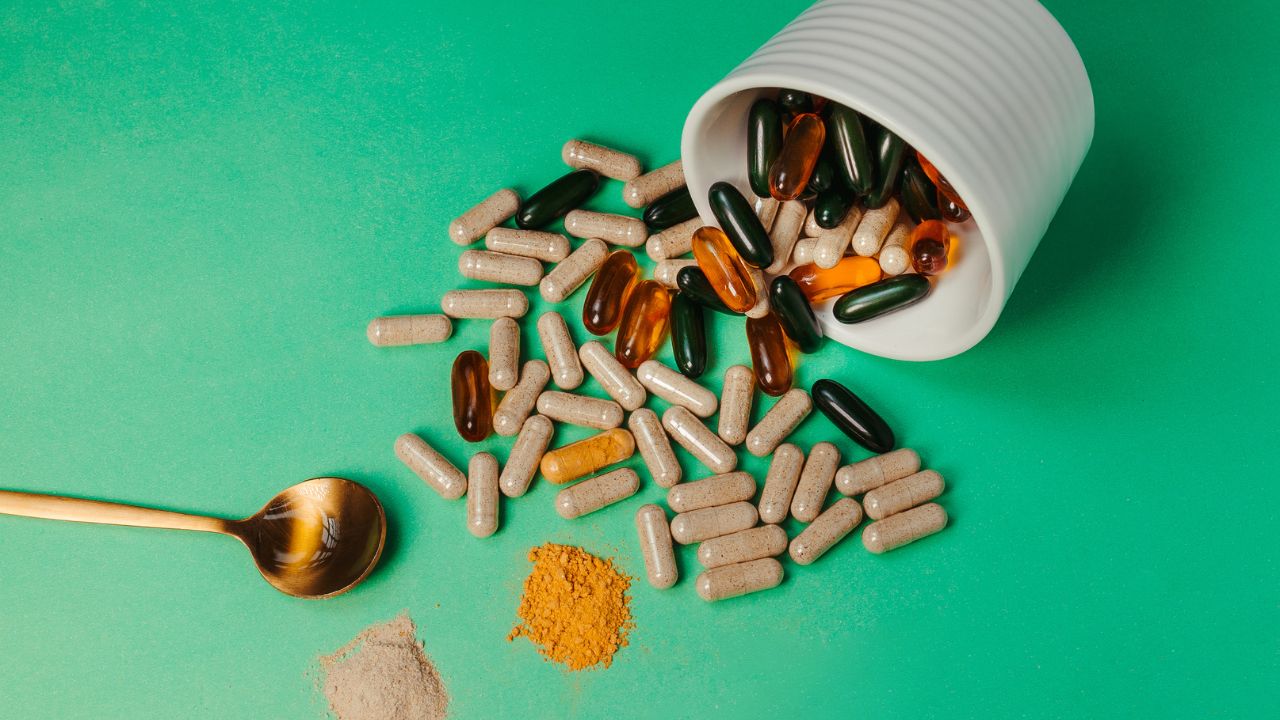











Be the first to react!