Researchers at Fred Hutch Cancer Center created antibodies from genetically modified mice that completely blocked infection by the Epstein-Barr virus — associated with cancers and multiple sclerosis
A study published in April 2026 in the journal Cell Reports Medicine revealed that scientists from the Fred Hutch Cancer Center in the United States developed monoclonal antibodies capable of completely preventing infection by the Epstein-Barr virus (EBV) in models with a human-like immune system.
The Epstein-Barr virus infects approximately 95% of the global population over a lifetime.
Most people carry the virus without knowing it.
-
Chinese ship nicknamed Bohai Sea Monster appears with four supports under the wings and draws the attention of military analysts in the Bohai Sea.
-
Human brains without bodies are kept “alive” in a laboratory in the USA to test drugs against Alzheimer’s and Parkinson’s while scientists face a debate about consciousness after death.
-
Tiny robots inspired by ants can build and demolish structures on their own in a Harvard experiment.
-
Mansion basement becomes archaeological site: engravings possibly made by enslaved people are found in a property over 260 years old in Ouro Preto
It is best known for causing mononucleosis, popularly called the “kissing disease.”
However, in more severe cases, it is associated with cancers such as Hodgkin lymphoma, Burkitt lymphoma, and nasopharyngeal cancer.
Risk of multiple sclerosis is 30 times higher in infected individuals
In addition to cancers, EBV is linked to severe autoimmune diseases.
A previous study from Harvard University showed that individuals infected with the Epstein-Barr virus have a 30 times higher risk of developing multiple sclerosis.
Despite this alarming association, to date, there are no specific vaccines or antivirals against EBV.
There are also no approved therapies to prevent infection.
Therefore, the discovery from Fred Hutch represents a significant advancement.

How the antibody blocked 100% of the infection
To develop the weapon against the virus, researchers used an innovative strategy.
They created genetically modified mice capable of producing human antibodies.
From these models, the team developed monoclonal antibodies that target two key proteins of the virus.
- gp350: protein responsible for allowing the virus to bind to host cells
- gp42: protein essential for the virus to penetrate immune cells
By blocking these two structures simultaneously, one of the antibodies completely prevented infection in mice with a humanized immune system.
The result was 100% prevention in the tests.

A virus that almost everyone carries unknowingly
The transmission of EBV goes beyond kissing.
The virus can be contracted through contaminated objects, blood transfusion, and even via the transplacental route — when the pregnant woman acquires the virus during pregnancy and passes it to the fetus through the placenta.
In most cases, infection occurs in childhood or adolescence and remains latent in the body for the rest of life.
The immune system keeps the virus under control but does not eliminate it.
That is why 95 out of every 100 adults worldwide carry EBV — usually without showing symptoms.
The problem arises when the immune system is compromised, such as in patients undergoing organ or bone marrow transplants.
Pathway to preventive therapy in transplant recipients
Researchers believe that the developed antibodies could be used as preventive treatment through infusions.
This approach would be especially useful for transplant patients, who receive immunosuppressive medications and become vulnerable to reactivation of EBV.
In these cases, the virus can emerge from latency and cause transplant-associated lymphomas.
With the new antibodies, it would be possible to neutralize the virus before it causes harm.
However, the therapy still needs to undergo clinical trial phases in humans to prove safety and efficacy.

Caveats: results are still from the laboratory
Despite the success in tests with mice, it is important to emphasize that the results are preclinical.
There is no information on the timeline for human testing or about possible side effects of the antibodies.
Additionally, the cost of any eventual therapy based on monoclonal antibodies is likely to be high, which could limit access in low-income countries.
However, the fact that for the first time an antibody managed to block 100% of EBV infections in models with a human immune system represents a milestone.
For a virus that 95% of humanity carries and that is linked to cancers and multiple sclerosis, any path to prevention is news worth paying attention to.

 Portuguese
Portuguese  English
English  Spanish
Spanish 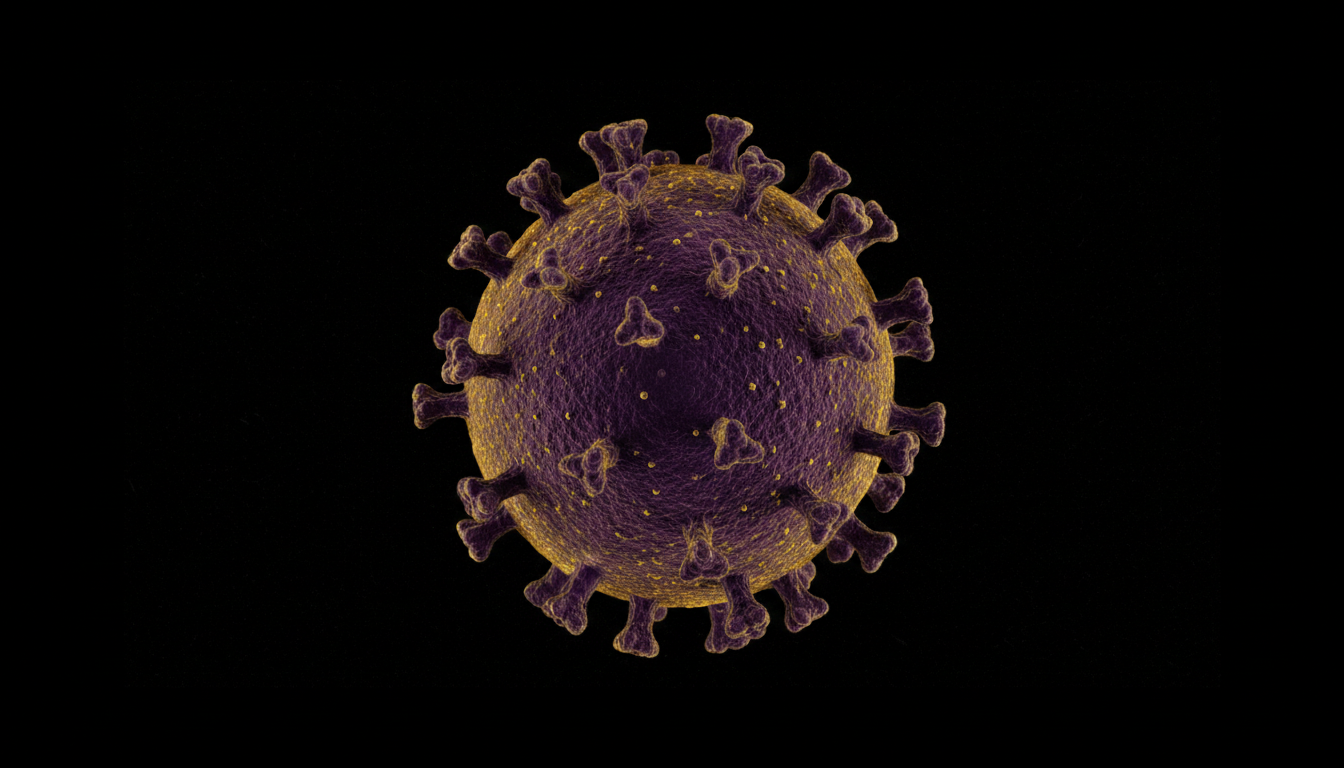










Be the first to react!