Process With Liquid Gallium Activated By Solar Light Reaches 12.9% Efficiency In Extracting Clean Hydrogen From Seawater And Freshwater, Proposes Reusable Chemical Cycle And Advances The Goal Of Expanding Sustainable Production On An Intermediate Scale
Researchers have developed a solar-powered process that utilizes liquid metals to produce clean hydrogen from seawater and freshwater, achieving a maximum efficiency of 12.9% and proposing a new sustainable pathway for energy generation.
The new method allows for the “collection” of hydrogen molecules from water, avoiding common limitations of current green hydrogen production processes. The proposal expands the possibilities for obtaining clean hydrogen based on renewable sources and widely available water.
According to lead author and PhD candidate Luis Campos, the process extracts sustainable hydrogen using only light as an energy source. He noted that the use of seawater increases access to the necessary resource for clean hydrogen production.
-
MIT study creates method to search for dark matter in gravitational waves and finds possible evidence in black hole signal
-
After 107 years at the bottom of the Atlantic, divers find the wreck of the Tampa, a World War I ship torpedoed with 131 people on board.
-
Scientists test 24 million routes and discover the most economical way to reach the moon using less fuel
-
Light-controlled switch uses only 4 femtojoules and brings photonic AI chips closer to a future with less heat and lower energy consumption
Dr. Francois Allioux stated that hydrogen can represent a clean energy solution and play a central role in an economy based on this resource. The project also highlights Australia’s strategic relevance in this international scenario.
The study was published in the journal Nature Communications. Senior researcher Professor Kourosh Kalantar-Zadeh from the School of Chemical and Biomolecular Engineering classified the results as a relevant demonstration of the natural chemistry of liquid metals applied to clean hydrogen production.
Clean Hydrogen And Initial Efficiency Of 12.9%
The team achieved a maximum efficiency of 12.9% in clean hydrogen production. Currently, researchers are working to improve this figure with the aim of commercializing the technology.
Kalantar-Zadeh compared the efficiency obtained with the historical trajectory of silicon-based solar cells. According to him, these cells started with 6% in the 1950s and did not exceed 10% until the 1990s.
For the team, the current rate is considered highly competitive as the first proof of concept. The goal now is to enhance the system’s performance while maintaining technical and economic viability.
The Role Of Liquid Gallium In The Circular Process
At the core of the technology is gallium, a low-melting metal that requires less energy to transition from solid to liquid state. The team has been exploring the chemical and technical limits of liquid metals for years.
The ability of gallium particles to absorb light was crucial for the development of the method. The particles are suspended in seawater or freshwater and activated under solar or artificial light.
Gallium reacts with water, transforming into gallium oxyhydroxide while releasing clean hydrogen. After hydrogen extraction, the oxyhydroxide can be reduced back to gallium and reused in the process.
This cycle characterizes the system as a circular chemical process. The reuse of gallium reduces material losses and reinforces the proposal for continuous production of clean hydrogen.
Surface Reaction And Properties Of The Metal
Liquid gallium has specific properties that favor the reaction. At room temperature, it appears solid, but when heated to body temperature, it turns into liquid metal puddles.
According to Campos, the surface of liquid gallium is chemically “non-stick,” making it difficult for most materials to adhere under normal conditions. However, when exposed to light in an aqueous medium, a surface reaction occurs.
In this condition, gallium oxidizes and gradually corrodes, forming pure hydrogen and gallium oxyhydroxide on the surface. This phenomenon allows the extraction of clean hydrogen at rates considered high by the team.
Kalantar-Zadeh stated that gallium had not been explored before as a means of producing hydrogen at high rates when in contact with water. According to him, it was a simple observation that had been overlooked.
Global Interest In Green Hydrogen
Hydrogen is regarded by industries and scientists as a relevant candidate for a sustainable energy source. Its use can help reduce greenhouse gas emissions.
So-called green hydrogen is produced from renewable sources. When burned, hydrogen does not produce pollutants, only water, while maintaining the capacity to generate high levels of energy.
Previous efforts focused on water photolysis, a process that separates hydrogen and oxygen atoms using electrolysis, photocatalysis, or plasma. These methods face multiple technical and economic obstacles.
Among the limitations are the need for purified water, high costs, or low hydrogen yields. The new method using liquid gallium avoids some of these barriers by allowing the use of seawater.
Next Steps And Expansion Of The Technology
According to Kalantar-Zadeh, there is a global need to commercialize highly efficient methods for producing green hydrogen. He stated that the developed process is efficient and easy to scale up.
The team is working to increase efficiency beyond the current 12.9%. The next goal is to build a mid-scale reactor to extract clean hydrogen in larger volumes.
Researchers are focused on refining the system and consolidating the circular process. The aim is to turn the proof of concept into practical applications for various sectors of the economy.
By integrating solar light, liquid gallium, and seawater, the method proposes a technical alternative for large-scale clean hydrogen production. The technology is still in the development phase, but it already shows results considered competitive by the team involved in the project.
This article was prepared based on information from the study published in the journal Nature Communications and statements from researchers Luis Campos, Dr. Francois Allioux, and Professor Kourosh Kalantar-Zadeh from the School of Chemical and Biomolecular Engineering.

 Portuguese
Portuguese  English
English  Spanish
Spanish 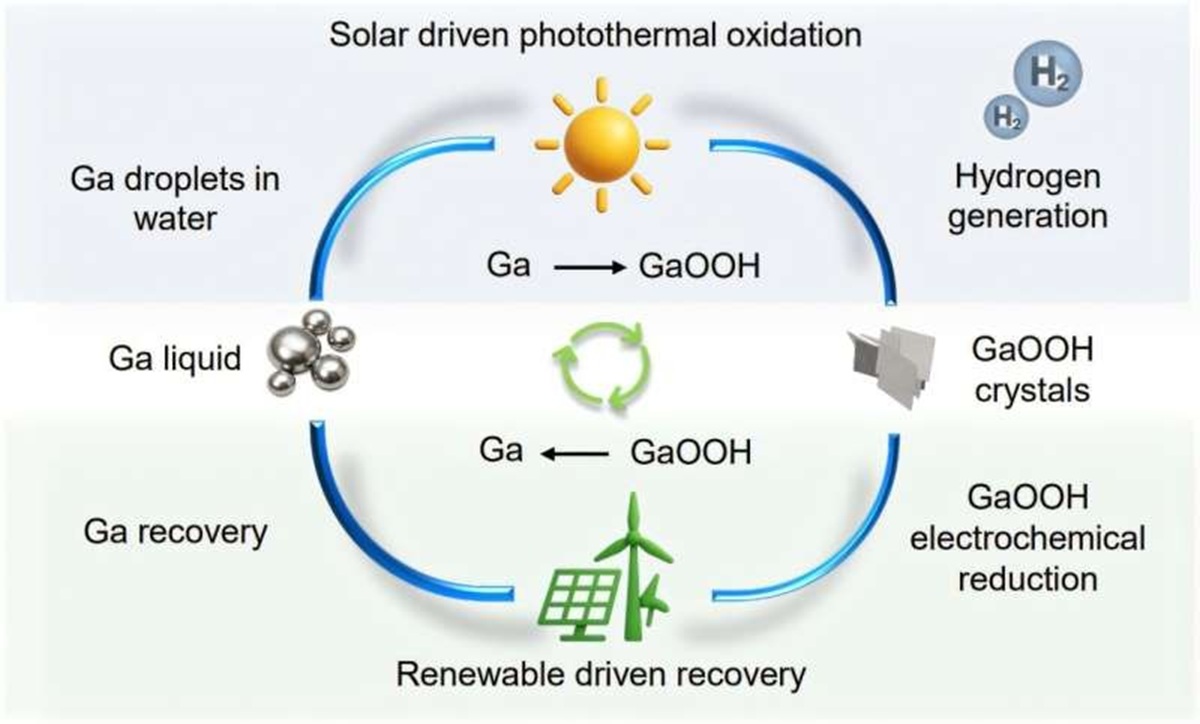








Be the first to react!