Researchers Propose A Middle Ground With Soft Electrolytes, Conciliating Performance And Manufacturing To Accelerate The Transition Of Solid-State Batteries. The Strategy Aims To Address Problematic Interfaces, Suppress Dendrites And Maintain High Ionic Conductivity Without Compromising Safety. In Focus, Hybrid And Porous Materials That Bring The Laboratory Closer To The Market.
Solid-state batteries promise more energy per volume, greater safety, and faster charging compared to liquid electrolyte lithium-ion cells. However, in practice, the direct leap from liquid to solid faces challenging technical barriers. Fragile interfaces, instability in air, and low ionic conductivity at room temperature still hinder widespread adoption.
In light of this impasse, the idea of a semi-solid electrolyte, or “soft electrolyte,” which functions as a structured gel, is gaining traction. According to researcher Yi-Cheng Deng from Wuhan University in China, advancing down this middle path could shorten the route to the ideal solid state. In 2026, Deng gathered evidence from various teams and outlined a roadmap of solutions that are already showing results.
The diagnosis compares the main solid electrolytes investigated so far. Oxide electrolytes are chemically stable but form poor interfaces with electrodes. Sulfide electrolytes conduct ions well but degrade in contact with air. Meanwhile, polymers are flexible and easy to process, but they conduct poorly under ambient conditions.
-
For the first time in history, a submarine cable will descend to four thousand meters deep under the ice of the North Pole to ensure that the internet between Europe and Asia no longer depends on conflict zones in the Middle East.
-
A British company has installed in the middle of the ocean the world’s first floating platform that generates electricity 24 hours a day from the temperature difference between the surface and the depths of the Atlantic, without relying on wind or sun.
-
The James Webb telescope spotted a planet 700 light-years from Earth with mornings full of sand clouds and nights with clear skies, the temperature difference between the two hemispheres reaches an impressive 170 degrees.
-
A former Hong Kong police officer has just become the first astronaut from her city to go to space. She embarked on the Shenzhou-23 mission alongside two other colleagues who will face muscle atrophy, radiation, and prolonged fatigue in orbit.
In this context, soft electrolytes attempt to combine the best of each world, balancing interfacial adaptation, mechanical robustness, and good ionic mobility. The proposal is pragmatic and focused on manufacturing viability, necessary to transition the technology from prototype to production line.
The Limitations Of Current Solid Electrolytes And The Impasse That Hinders Large-Scale Adoption
According to Deng, the chemical stability of oxide electrolytes does not compensate for the low interface quality with electrodes, which impedes ion passage and increases internal resistance. This incompatibility reduces lifespan and increases energy losses.
In the case of sulfides, the electrochemical performance is promising, but degradation in air complicates manufacturing and handling outside controlled environments. This raises costs, increases risks, and limits industrial scale.
Polymers stand out for their flexibility and ease of processing, but insufficient ionic conductivity at room temperature requires additives or heating, compromising efficiency, cost, and ease of use.
The Middle Path With Soft Semi-Solid Electrolytes And The Two Strategies That Are Already Showing Results
The first strategy described by Deng is the rigid-flexible synergy composites. It combines a flexible phase — such as polymers, ionic liquids, or plastic crystals — with rigid inorganic fillers, typically nanoscale oxides. This architecture seeks intimate contact with the electrode without sacrificing mechanical strength.
In this approach, the rigid part acts as a “skeleton” that helps to suppress dendrites and guide lithium ion transport, while the flexible phase improves interface and processability. The result is a more uniform, stable, and production-compatible electrolyte.
The second front is the lithium desolvation mechanism, which employs porous materials such as metal-organic frameworks (MOFs) and covalent organic frameworks (COFs). Their nanometer-scale channels function as “cages” that immobilize solvent molecules.
By regulating the solvation structure of lithium ions, these “molecular cages” tend to broaden the electrochemical stability window and increase compatibility with metallic lithium. In practice, this reduces unwanted reactions and favors longer cycles.
What Has Been Achieved And Why This Could Accelerate The Transition From The Laboratory To The Market
According to Deng, soft electrolytes offer a balanced portfolio of properties: higher ionic conductivity, robust mechanical integrity, excellent interfacial adaptation, and better processability. These factors align directly with real-world battery requirements.
The team is optimistic about the pace of advancements. In the researcher’s words, flexible solid-state electrolytes are poised to accelerate the transition from the laboratory to the market, supporting safer and denser systems for transportation and renewable energy integration.
Engineering And Manufacturing Challenges That Need To Be Solved To Scale The Technology
Despite the progress, there are intrinsic bottlenecks. One of them is achieving homogeneous dispersion of components within the electrolyte, avoiding clumping that creates mechanical weak points and preferential pathways for dendrites.
Another challenge is the stabilization of multiphasic interfaces — solid, gel, and liquid — over many cycles. Maintaining these chemical and mechanical boundaries is crucial to prevent premature degradation and performance losses.
On a practical level, the industry needs reproducible methods to scale production of soft electrolytes. Bench-scale processes do not always easily translate to high-throughput lines, with quality control and competitive costs.
The solution requires a coordinated effort among materials, cell architecture, and industrial processes. Adjustments to stacking design, contact pressure, and surface treatments can be decisive in ensuring stable interfaces.
With clear scalability, safety, and cost goals, soft electrolytes could solidify a transition pathway while ideal solid electrolytes continue to be developed. The balance between performance and manufacturing is the axis of this strategy.
Realistic Next Steps And How To Measure Success In Soft Solid-State Batteries
Advancements depend on standardized protocols to assess lifespan, safety, and compatibility with metallic lithium, as well as metrics for dendrite suppression. Reproducible tests will facilitate fair comparisons and engineering decisions.
In the same vein, industrial pilots must validate processability and quality at scale, from moisture control to deposition and lamination routes. Success will come when electrochemical performance and manufacturing viability progress together.
What do you think about the bet on soft electrolytes as an intermediate step toward full solid state? Does it accelerate the arrival of safer and more autonomous electric cars, or creates a “middle ground” that might delay the ideal? Leave your comment and say whether you would prioritize maximum performance now or manufacturing robustness with gradual gains. The technical debate helps to separate hype from a viable path.

 Portuguese
Portuguese  English
English  Spanish
Spanish 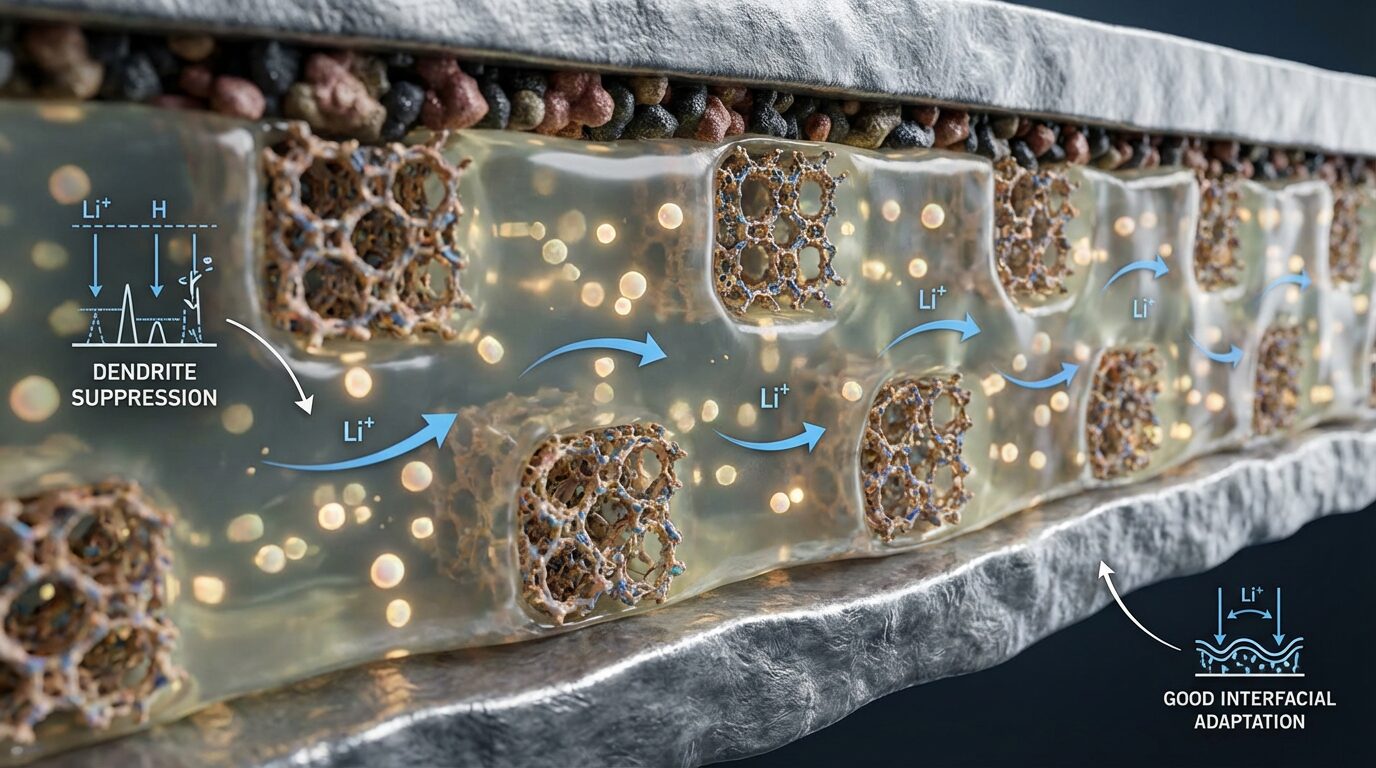








Be the first to react!