Caltech Researchers And Southern California University Created Biocompatible Bubble Robots That Move Using Body Chemical Reactions, Can Reach Tumor Regions And Release The Therapeutic Load On Command.
Researchers from Southern California University create enzyme-based microrobots that seem like something out of a movie inside the human body, but there is a problem: most ideas look good in slides and are boring in real life. Either they are too complex, or they cost too much to manufacture, or they become difficult to control once they enter living tissue, where everything is viscous, tight, and unpredictable.
The insight of this new work was to remove excessive engineering from the equation and bet on the bare minimum that works: a bubble.
Yes, a microbubble, one of those that have been used for years in imaging exams. The team took this “common object” from medicine and transformed it into a microrobot with very straightforward functions: carry a drug, move to the tumor, and release the load at the right moment.
-
Scientists in Saudi Arabia create an “electricity-free refrigerator” that uses a chemical reaction to lower the temperature from 25°C to 3.6°C in 20 minutes and then uses the sun itself to restart the system without a compressor, outlet, or generator.
-
MIT study creates method to search for dark matter in gravitational waves and finds possible evidence in black hole signal
-
After 107 years at the bottom of the Atlantic, divers find the wreck of the Tampa, a World War I ship torpedoed with 131 people on board.
-
Scientists test 24 million routes and discover the most economical way to reach the moon using less fuel
The result draws attention precisely because it does not rely on a structure full of parts. Instead of 3D printing, hydrogel, and cleanroom manufacturing, the robot emerges as a microbubble coated with proteins, with chemical adjustments on the surface to couple enzymes, drugs, and functional particles. This allows for the rapid production of many similar units at a lower cost, a point that often kills microrobotics projects when they try to leave the lab.
Why A Bubble Can Be A Robot And What It Gains From It
The microbubble has two huge advantages. The first is biocompatibility because it has been used in the human body for quite some time in imaging applications.
The second is that it is, at the same time, a capsule and a mechanism. It can carry things on its surface and can also be broken in a controlled manner, which becomes a clean way of releasing on demand.
The team created protein capsules around the bubbles and, from there, began to “equip” these bubbles as one would add functions to a Swiss Army knife.
The surface chemistry became the control panel. You can couple the enzyme that generates movement, attach the anticancer drug, and add particles that facilitate tracking or direction.
What makes everything more interesting is that the system allows for two modes of operation. One more guided and another more autonomous. And this aligns with the real world because not every clinical application will want the same level of external control.
How These Microbubbles Manage To Move Without A Motor And Without A Battery
This is where the part comes in that makes the reader’s eyes widen. The movement does not come from a propeller, does not come from a battery, does not come from a motor. It comes from an enzyme.
One of the versions uses urease, bound to the surface of the bubble. This enzyme reacts with urea, a waste product naturally present in the body, generating ammonia and carbon dioxide.
However, the distribution of the enzyme on the bubble is not perfectly uniform, so the reaction products accumulate more on one side. This creates a chemical push that propels the bubble forward.
In practice, the bubble becomes a self-propelling microrobot that moves by leveraging the body’s environment. It is not Formula 1 speed, but it does not need to be. The goal here is to get to the right place efficiently, not to run a marathon.
Additionally, one version includes magnetic nanoparticles, opening the possibility of orientation by external magnets, with ultrasound tracking.
The other version seeks true autonomy, using a second enzyme to respond to chemical signals typical of tumors.
When The Tumor Becomes A “Smell” And The Robot Follows The Gradient
tumors and inflamed tissues tend to have higher concentrations of certain compounds. One of them is hydrogen peroxide. By adding catalase to the system, the researchers created a mechanism that responds to this chemical gradient.
In other words, the microrobot starts to “sense” where there is more peroxide and moves in that direction, as if it is following a trail. This type of orientation is known as chemotactic guidance.
The important detail is that, in this mode, the robot does not need a person guiding it in real time or a heavy external control scheme to find the target. It uses the tumor’s environment as its compass.
This is where the project becomes dangerously interesting because it suggests a more precise delivery route with a simple structure. And simplicity is often what allows for scaling.
The researchers did not stop at laboratory talk. They tested the concept in an animal model and put numbers on the table, comparing the delivery performance with conventional therapy.
The full description of the method, the two versions of the microrobot, and the results of these experiments appear in a study published in Nature Nanotechnology, where you can clearly see what was actually demonstrated and what still depends on validation in future stages.
The Final Trigger Is Ultrasound And The Release Happens At Once
Getting to the tumor is only half the problem. The other half is getting the drug to actually enter the tumor tissue. Many systems release slowly, and this does not always solve the problem because penetration can be limited.
Here, the strategy was to use focused ultrasound to break the microbubble when it reaches the target. The collapse is rapid, releasing the load all at once and creating a mechanical effect that helps the drug penetrate more deeply. It is a type of delivery that avoids the standard of “dripping slowly” and bets on controlled impact.
In experiments with mice with bladder cancer, the approach led to a significant reduction in tumor weight over 21 days compared to animals treated only with the drug, suggesting that the delivery method made a difference and not just the drug itself.
The point that makes this system seem more ready for the world is the combination of four things in the same package: biocompatibility, movement, targeting capability, and on-demand release. All this without becoming an expensive robot that is impossible to manufacture.

 Portuguese
Portuguese  English
English  Spanish
Spanish 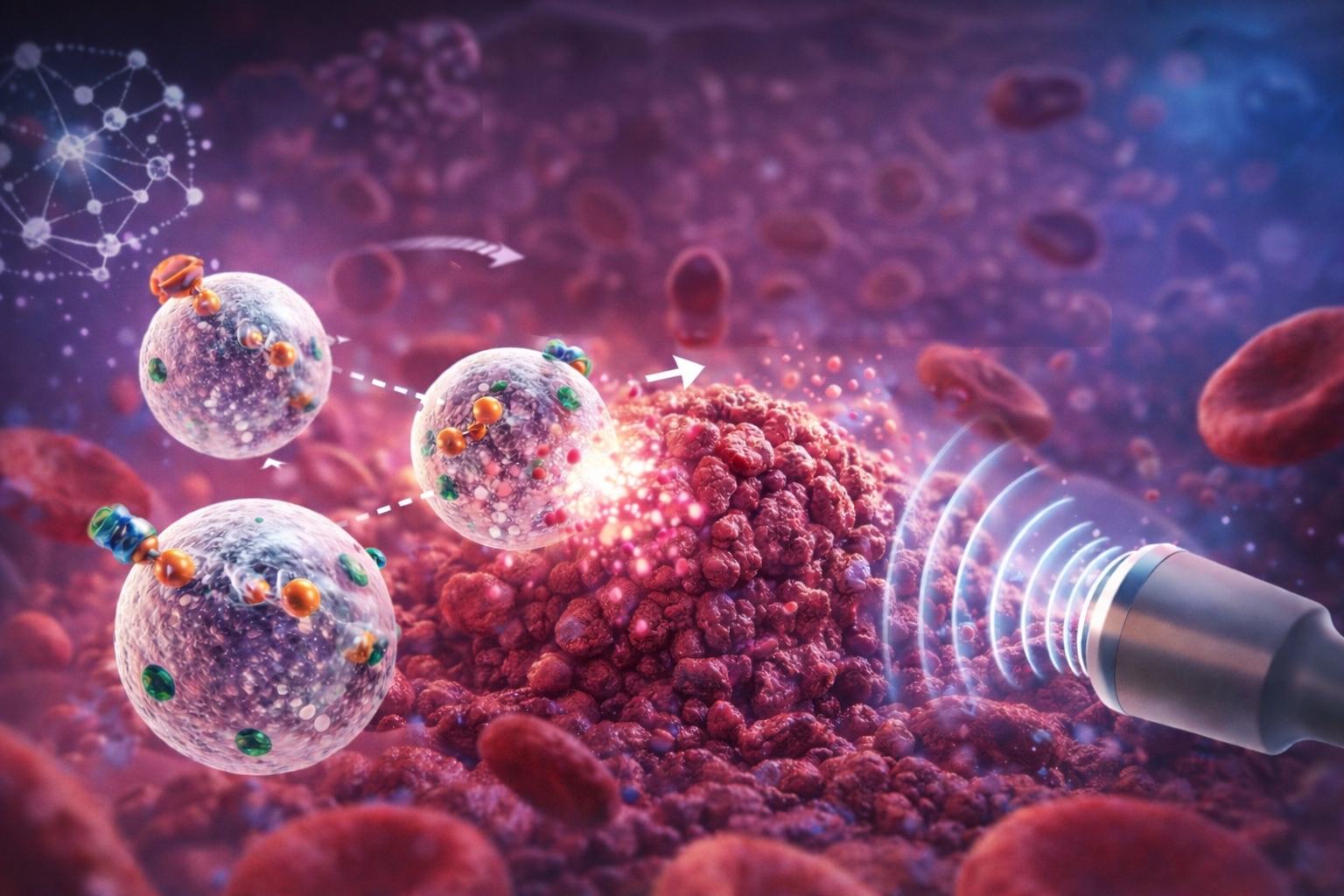









-

1 person reacted to this.