A study in Nature Microbiology reveals how ‘kamikaze’ bacteria sacrifice themselves for the group to disseminate resistance to drugs. Scientists are now investigating how to block this mechanism and contain the propagation of superbacteria.
A bacterium that destroys itself to save others. What seems like a science fiction script has been confirmed by researchers at the John Innes Centre in the UK, in a study published in the journal Nature Microbiology on April 16.
The discovery shows that certain microorganisms are capable of intentionally rupturing themselves, releasing particles loaded with genetic material — including antibiotic resistance genes — that are then absorbed by neighboring bacteria.
The mechanism, more sophisticated than previously thought, highlights one of the most concerning processes in modern microbiology: horizontal gene transfer among bacteria.
-
A large part of the Netherlands lies below sea level and remains dry only because an invisible machine of dikes, pumps, and giant gates works nonstop, a system born from the tragedy of 1953, when the North Sea invaded the country and killed 1,836 people.
-
The largest bridge in Finland has just been inaugurated and cars simply cannot pass on it; only trams, bicycles, and pedestrians are allowed to cross the 1.2 km giant with a 135-meter pillar over the Baltic Sea.
-
Larger than Belgium and nestled at the mouth of the Amazon, Marajó Island is a giant that Brazil has forgotten, surrounded by water and energy on all sides, yet still isolated, poor, and dependent on diesel brought by barge.
-
Weighing as much as nine nuclear aircraft carriers, the Pioneering Spirit is the largest ship ever built and was made for a single task that no other machine in the world can accomplish: removing entire oil platforms from the seabed of the North Sea in one go.
What are ‘kamikaze’ bacteria and why do they matter
The term “kamikaze” may sound dramatic, but it accurately describes the observed behavior: the bacterium self-destructs for the benefit of the collective. This type of behavior is not new in nature — human cells also undergo programmed death processes to protect the organism. What is surprising here is the sophistication of the bacterial mechanism and, above all, what it carries with it when it ruptures.
When a bacterium collapses in this process, it does not simply disappear. It releases structures called gene transfer agents (GTAs) — particles morphologically similar to viruses, but that function as genetic messengers. These particles capture DNA fragments from the destroyed cell and transport them to other surrounding bacteria.
The practical result is straightforward: advantageous genes — such as those that confer antibiotic resistance — can spread quickly and efficiently throughout an entire population of microorganisms, even without direct reproduction.
Evolutionarily domesticated viruses: the unlikely origin of GTAs
One of the most revealing aspects of the study is the origin of GTAs. Scientists identified that these particles are, in fact, remnants of ancient viruses that infected bacteria long ago and, over the course of evolution, were “domesticated” by the microorganisms themselves.
Instead of destroying host cells — as common viruses do — these elements were repurposed as tools for genetic communication. It is a complete reversal of function: what was once a threat has become an ally.
This type of evolutionary repurposing is not uncommon in biology. The human genome, for example, carries viral-origin sequences that today perform essential regulatory functions. In the case of ‘kamikaze’ bacteria, however, the reuse of the viral mechanism directly serves the collective survival of the species — and, consequently, the spread of traits that hinder medical treatments.
LypABC: the three-gene switch that makes the cell explode
Until the publication of this study, one point remained unclear: how do bacteria exactly control when and how this self-destruction process occurs? The research identified the answer in a set of three genes called LypABC.
This trio functions as a molecular switch. When activated, it causes cell lysis — that is, it makes the bacterial membrane rupture and release the GTAs into the external environment. The balance is delicate:
- Without proper activation, the process simply does not occur
- With deregulated activation, the system becomes toxic and can destroy cells excessively, compromising the colony itself
Interestingly, the LypABC mechanism bears structural similarity to bacterial immune systems typically used to defend against viral invaders. The difference is that, in this case, the system has been reconfigured to do the opposite: open the cell and release its genetic content, rather than protect it. This duality of function — defense and self-sacrifice using the same molecular machinery — points to an evolutionary plasticity that scientists are still learning to map.
Horizontal gene transfer: the silent engine of antibiotic resistance
To understand the real impact of the discovery, it is necessary to comprehend the central process it involves. Unlike vertical inheritance — where genes pass from parent to child during reproduction — horizontal gene transfer allows bacteria to exchange genetic material directly with each other, even without kinship.
It’s as if an organism could acquire, in a matter of hours, a skill that would take generations to arise through spontaneous mutation. The main ways this process occurs in bacteria include:
- Transformation: direct absorption of DNA fragments from the environment
- Conjugation: transfer by physical contact between two cells
- Transduction: virus-mediated transport — or, as in this case, by particles that behave like viruses
What the new study reveals is that ‘kamikaze’ bacteria make the third mechanism even more efficient. By deliberately destroying themselves, they maximize the amount of GTAs released, increasing the likelihood that other cells will absorb the resistance genes. In practice, a single bacterium can, by sacrificing itself, contribute to making others equally resistant to one or more antibiotics.
The human cost of antimicrobial resistance is already measurable
The World Health Organization and The Lancet classify antimicrobial resistance as one of the ten greatest threats to global public health. The estimates are concrete: infections by antibiotic-resistant bacteria are already directly responsible for more than 1.2 million deaths per year worldwide.
In this context, understanding the mechanisms that accelerate the spread of resistance genes is not just scientific curiosity — it is an urgent clinical necessity. The identification of the LypABC system as a trigger for kamikaze behavior opens a promising research avenue.
If it is possible to block this molecular switch, it may be feasible to reduce the speed at which resistant bacteria spread, not only among patients but within the organism itself and in high-risk environments.

Blocking the molecular trigger could be the next step against superbacteria
The scientists involved in the study point out that the next step is to accurately understand how the LypABC system is activated under natural conditions. What environmental or internal signals trigger the sequence? Is there any natural inhibitor? Is it possible to create a compound that interferes with this process without affecting other bacterial functions?
These questions remain unanswered. But the fact that the mechanism has been identified and described in molecular detail represents a concrete advance. In applied science, accurately naming the mechanism is the first step to neutralizing it.
The discovery of ‘kamikaze’ bacteria redefines part of what was understood about microbial cooperation. It shows that the bacterial world operates with more elaborate collective strategies than previously thought — and that understanding how bacteria cooperate may paradoxically be the key to preventing that cooperation from working against us.

 Portuguese
Portuguese  English
English  Spanish
Spanish 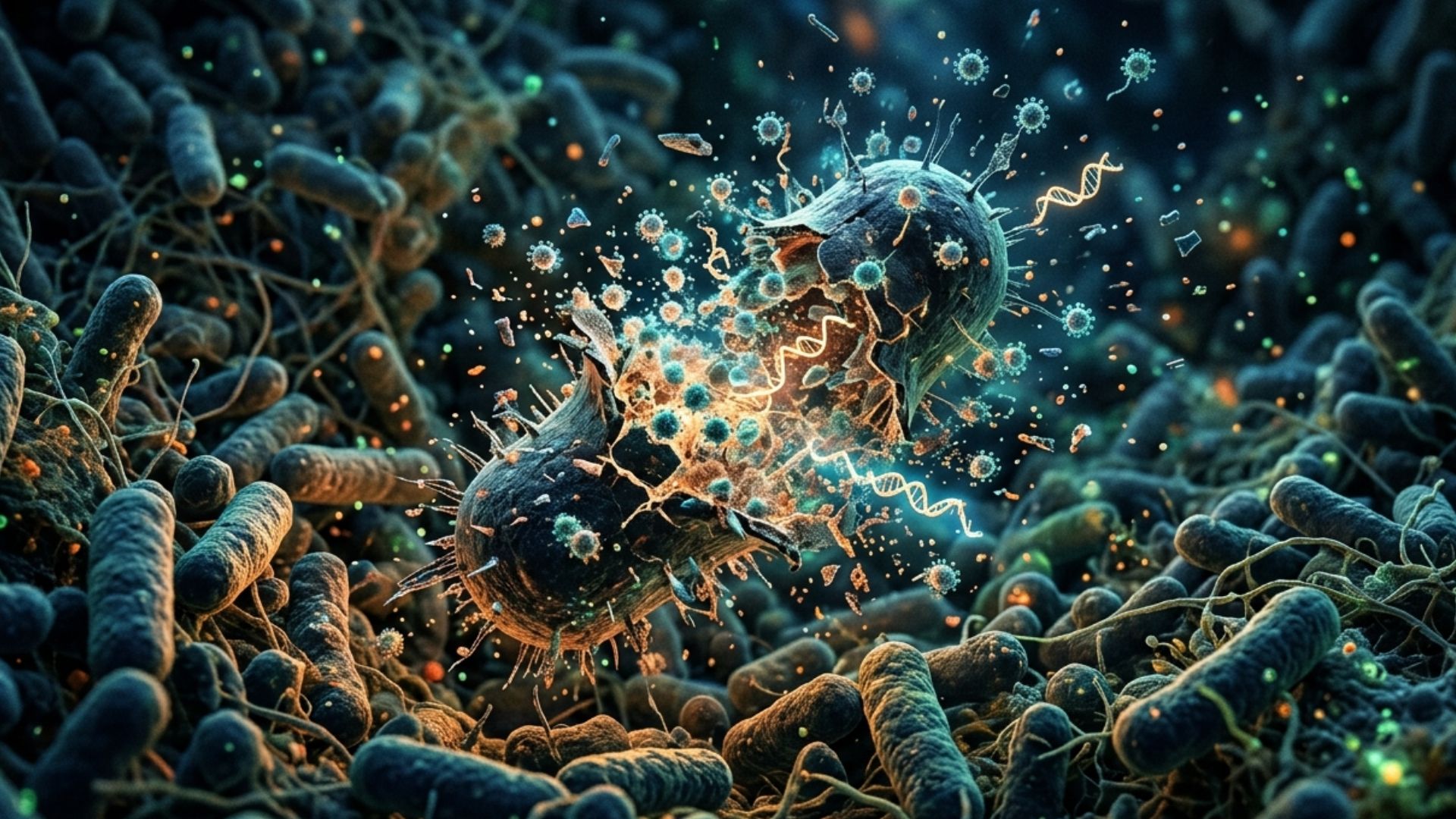













-

1 person reacted to this.